THE UNITED REPUBLIC OF TANZANIA PRESIDENT'S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT

MBEYA CITY ACADEMIC COMMITTEE
FORM FOUR PRE MOCK EXAMINATION
CODE: 032 CHEMISTRY
(For Both School and Private Candidates)
TIME: 3:00 HOURS APRIL 2025
INSTRUCTIONS
- This paper consists of three (3) sections A, B and C with a total of eleven(11) questions.
- Answer ALL questions in sections A and B and only two (2) questions in section C.
- All writings should be in blue or black ink, EXCEPT for diagrams which must be in pencil.
- Cellular phones and any unauthorized materials are not allowed in the examination room.
- Non-programmable calculators may be used Where calculations are involved, show your work clearly.
- Write your Examination number on every page of your answer sheet(s).
- You may use the following atomic masses and constants:
- Atomic masses:
- H = 1, C = 12, O = 16, Na = 23, S = 32, Cl = 35.5, Ca = 40, Cu = 64, Ag = 108, Fe = 56.
-Constants:
- Avogadro’s number, NA = 6.02 x 1023
- GMV at STP = 22.4 dm3
- 1 Faraday = 96500 Coulombs
- 1 L = 1 dm3 = 1000 cm3
SECTION A: (16 Marks)
Answer ALL questions in this section
1. For each of the following items (i) - (x), choose the most correct answer from among the given alternatives and write its letter beside the item number.
(i) Which name is given to the process of dissolving sodium chloride in water?
- Precipitation process
- Neutralization process
- Hydration process
- Dissolution process
- Dehydration process
(ii) Carbon IV oxide gas contains Oxygen but it is used to extinguish fire. Why?
- Because it is wet
- Because it is denser than air
- Because it is less dense than air.
- Because it burns at high temperature
- Because it is not inhaled.
(iii)When a mixture of liquids is boiled for separation of the constituents, the constituent that is separated last has the:
- Lowest boiling point.
- Highest boiling point.
- Moderate boiling point.
- Lowest and highest boiling point.
- Highest and moderate boiling point.
(iv)Aluminium dose not react with water and does not corrode much in air because:
- It is below Hydrogen in the reactivity series.
- It forms a stable carbonate which prevents reactions.
- The metal is covered with a protective coating of an oxide.
- It is very stable to every reaction.
- Aluminium ions do not react with water.
(v) When a burning fuel produces blue flame colour it means there is:
- adequate supply of Oxygen with production of soot.
- inadequate supply of Oxygen without production of soot.
- Inadequate supply of Oxygen with production of soot.
- Adequate supply of Oxygen with less production of heat.
- Adequate supply of Oxygen with production of more heat.
(vi ) Water has a relative high specific heat capacity. This property makes it suitable for use as:
- Solvent
- Cleaning
- Coolant
- Absorbent
- Catalyst
(vii) When the gases Sulphur dioxide and Hydrogen sulphide mix in the presence of water, the reaction is: SO2(g) + 2H2S(g) ![]() 2H2O(l) + 3S(s).
2H2O(l) + 3S(s).
Here, Hydrogen sulphide is acting as:
- An oxidizing agent
- A reducing agent
- A catalyst
- A dehydrating agent
- An oxidant
(Viii) Which of the following sets of laboratory apparatus are used to measure volume of water?
- Thistle funnel, separation funnel and beaker
- Burrete, pipette and measuring cylinder
- Crucible, Kipp’s apparatus and volumetric flask.
- Volumetric flask, distillation flask and test tube.
- Test tube, beaker and gas jar
(ix)Which of the following sets of processes uses a gas that ignites a “POP” sound when a lighted splint is passed through it?
- Balloon filling, welding and diving.
- Balloon filling, fuel rocket and diving.
- Hardening oil, balloon and welding.
- Hardening oil, balloon and diving.
- Fueling rocket, diving and welding.
(x) Which of the following transforms a mechanical energy to an electric energy?
- Hydroelectric power plant
- Solar panel
- An electric cooker
- An electric motor
- A motorcycle wheel
2. Match the chemical composition of fire extinguisher in List A with the corresponding extinguisher in List B by writing the letter of the correct response beside the item number in the answer booklet provided.
| LIST A | LIST B |
| (i) The extinguisher composed of bromochlorodifluoro-methane. (ii) The extinguisher composed of potassium acetate. (iii) The extinguisher composed of monoammonium phosphate with a nitrogen carrier. (iv) The extinguisher composed of fine sodium bicarbonate powder pressurized by nitrogen. (v) The extinguisher composed of proteins and fluoro-proteins. (vi) The extinguisher composed of tap water pressurized by air. |
|
SECTION B: (54 Marks)
Answer all questions in this section
3 (a) The pH indicators are used in various ways. Briefly explain the areas where it is used. (Four points) (04 marks)
(b) The following results were obtained in an experiment to find the molecular formula of magnesium oxide when magnesium burnt in air.
Mass of crucible = 12.5g
Mass of crucible + magnesium = 14.9g
Mass of crucible + magnesium oxide = 16.5g
(i) What mass of magnesium was used in the experiment?
(ii) How many moles of magnesium atoms were used in the experiment?
(iii) What mass of oxygen combined with magnesium?
(iv) How many moles of oxygen combined with magnesium?
(v) Use the results in (b) (i)-(iv) to write the molecular formula of magnesium oxide. (5marks)
4.(a) Give good reason(s) for each of the following (answer according to the question demand).
- Natural gas is so popular in heating and cooking at homes. (one point)
- Nuclear energy is not a suitable source of energy. (one point)
- Coal and Petroleum are non-renewable sources. (One point) (3marks)
(b) A chemical substance L has a density of 1 g/cm3 at 4 0C, it is neutral to litmus paper, it freezes at at 0 0C and boils at 100 0C
- Specify the name and the chemical formula of substance L
- How can substance L be confirmed in the laboratory? Describe two confirming methods and provide its positive observations.
- State two main physical properties of substance L and show the usefulness of each property in our daily life. ( 6marks)
5 .(a) Souring of milk and melting of ice both are changes of substance. Based on the properties of their changes distinguish them by giving four (4) points. (04 marks)
(b) ) Explain each of the following observations.
- Why 2-Methylpropane has a lower boiling point than butane although both have the same molecular mass.
- Why ignition sources such as smoking are not allowed at petrol stations. (5marks)
6. (a) A Form Three student conducted an experiment to prepare a gas in the laboratory by decomposing a certain compound using electricity. She allowed a steady current to flow through the solution for 3 hours at S.T.P. If the volume of the gas obtained was 4.12 dm3 and the gas relighted a glowing splint.
- Name the gas that was produced. (01 mark)
- Calculate the electric current that was flowing in the solution. ( 3marks )
(b) Metals are extracted from their ores. Using two important metals in the table below, identify the substance(s) apart from their chief ores which are added during the extraction process and finally, suggest their functions.
| Name of the metal to be extracted | Substance(s) added during extraction | Function(s) of the substance(s) |
| Sodium | ||
| Iron |
5marks
7. (a)(i)A cough syrup contains 5.0 % of ethyl alcohol (C2H5OH) by mass. If the density of the alcohol is 0.993 g/cm3, what is the molarity of the alcohol in the syrup. (03 marks)
(ii) If a sick child was given 2.5 mls of ethyl alcohol, then determine the mass taken by the child.(02 marks)
(b) The part of Periodic table is shown below. The letters are not the usual symbols for elements.

- Which elements have the same electronic arrangement as the stable ion of F+ and A-? (01 mark)
- Write two properties of the compound (bond) formed between D and A. (01 mark)
- Element C and G were complete burnt in air. Write down equations to show the
- Combustion of each of the element and the type of oxide formed. (02 marks)
8. (a) Michael was performing experiment in the chemistry laboratory but finally he failed to success in obtaining required result. In his experiment. As a form four student show skills Michael should have in order to obtain the required result from his experiment. (6) points. (6marks)
(b) Suggest three sources of heat that can be used in a chemistry laboratory apart from Bunsen burner (3marks.)
SECTION C: (30 Marks)
Answer only two (2) questions in this section
9. (a) Molarity of solutions is always affected by change in temperature. Support this statement with a mathematical expression. (02 marks)
(b) 0.2 g of impure iron sample reacted with 27 cm3 of 0.3 M HCl solution to form iron (III) chloride and hydrogen gas. Calculate the percentage purity of iron. (07 marks)
(c) Sulphuric acid is available as a stock solution with the following information:
Purity = 98 %, Density = 1.84 g/cm3, Molecular weight = 98.08 g/mol.
- What volume of the stock solution should be used to prepare 100 cm3 of 1 M sulphuric acid solution? (04 marks)
- What volume of distilled water should added in the stock solution in order to prepare a 100 cm3 of 1 M sulphuric acid solution? (02 marks)
10. The agricultural officer at Manga ward conducted a meeting with all farmers in that ward to discuss the activities which cause the loss in soil fertility in their land. Describe any six possible ways which were motioned by the officer at that meeting.
11. Form three students were Conducted the practical in the laboratory, the practical involved reagent D and E, after the reaction the results were Universal solvent and salt only.From the practical Conducted identify the reaction and explain in details about five applications of identified reaction in our daily life
FORM FOUR CHEMISTRY EXAM SERIES 247
FORM FOUR CHEMISTRY EXAM SERIES 247
THE UNITED REPUBLIC OF TANZANIA PRESIDENT’S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
DODOMA REGION
FORM FOUR MOCK- EXAMINATION
032/1 CHEMISTRY 1
Time: 3 Hours August, 2023
Instructions
- This paper consists of section A, B and C with total of eleven (11) questions
- Answer all questions in section A and B and two (2) question from section C
- Cellular phones and any unauthorised material are not allowed in the examination room
- Write your Examination Number on every page of your answer booklet(s)
- Non programmable calculator may be used
- The following constant may be used Atomic masse:
- H=1, C=12, N=14, O=16, Na=23, S= 32, Ca=40, Cl= 35.5, Cu= 64 and Zn=65 Avogadro’s number = 6.02 x 1023
- GMV at s.t.p = 22. 4 dm3
- 1 faraday = 96,500 coulombs
- Standard pressure = 760 mm Hg
- Starndard tempreture = 273K
- 1 litre = 1 dm3= 1000cm3
SECTION A: (16 marks)
Answer all questions in this section
1. For each of the items (i)-(x) choose the correct answer from given alternatives and write its letter beside item number in the answer booklet provided
i. Which of the following is not a use of a solvent?
- Bleaching agent
- Greasing.
- Stain removal.
- Cleaning
- Universal solvent
ii. In which step of scientific procedures does the hypothesis is either proved or disproved?
- Data collection and analysis.
- Experimentation.
- Data interpretation
- Formulation of hypothesis.
- Conclusion
iii) Which of the following groups consist of home Care products?
- Yeast, plastic and disinfectant.
- Clothes, soap and stone.
- Air freshener, detergent and antiseptic.
- Petrol, air freshener and paints.
- Air freshener, detergent and disinfectant
iv. An electric current of 0.2A was passed through an electrolyte for 16.67minutes. The quantity of electricity passed is;
- 200.04cuolombs
- 2000.004cuolombs
- 1000cuolombs
- 0.254cuolombs
- 0.00789culombs
v. What are the main components of crude oil?
- Gases, liquids and solids.
- Gases, water and diesel.
- Mineral, salts and oils
- Gasoline, hydrogen and milk.
- Methane only
vi. What will happen when sulphur dioxide is mixed with potassium dichromate (iv) solution in Presence of sulphuric acid?
- Sulphur will be formed.
- Green chromate (III) sulphate will be formed
- Blue water will be formed.
- Sulphur dioxide gas will be formed
- Orange potassium sulphate will be formed
vii. Which of the following reaction is a most convenient method of producing sulphur dioxide during contact process
- Iron (II) sulphide react with oxygen to form sulphur dioxide and iron(III) oxide
- Zinc sulphide reacts with oxygen to form sulphur dioxide and zinc oxide
- Sulphur reacts with oxygen to form sulphur dioxide
- Oxygen reacts with hydrogen to form water
- Sulphur dioxide reacts with oxygen to form sulphur trioxide
viii. The copper (II) oxide reacts with hydrogen gas to form copper metal and water. What will be the mass of reduced element?
- 4g.
- 64g.
- 18g.
- 80g.
- 40g
ix. During the steam reforming method in industrial preparation of hydrogen, the steam reacts with what compound to produce hydrogen gas?
- Water.
- Carbon monoxide.
- Methane.
- Sulphur dioxide.
- Oxide
x. What will be the simplest test for the hardness of water?
- Shaking water with chalk.
- Mixing water with soap less detergent.
- Formation of scum.
- Shaking water with soap solution.
- Formation of dolomite
2. Match the colour formed in LIST A with the corresponding metal burning in air in LIST B
| LIST A | LIST B |
|
|
SECTION B (54marks)
Answer all questions in this section
3. (a) Students are advised to use a non- luminous flame for heating in the laboratory
(i) Explain how a Bunsen burner produces a non- luminous flame
(ii) Give a reason as to why advice above given to students.
(iii) What are the functions of the air holes and barrel in the Bunsen burner?
(b)Why hydrogen peroxide preferred to potassium chlorate during preparation of oxygen gas?
(c) Why iron is not usually recommended in construction of steam pipes and boilers?
(d) What would happen to a well stoppered bottle full of water left in deep freezer over night? Why does this happen.
4. People of Uweto region were complaining about the water they are using in everyday activities that contain different contaminants and pollutants like bad smell, coloured matter, floating materials and other different solid substances. The Uweto people wish their water to be treated and purified but they do not know what to do in treating and purifying water. As a Chemist help Uweto people on how to treat and purify their urban water
5. (a) Explain why carboxylic acid has high melting point and boiling point compared to their corresponding alcohol?
(b) With help of chemical equation, state two chemical properties of carboxylic acid.
(c) Explain why does fractional distillation of crude oil gives different products
(d) (i) Define the term hydrocarbons.
(ii) Write down the molecular structure and I.U.P.A.C names of the isomers whose molecular formula is C4H10.
6. (a) In electrolytic production of hydrogen gas, dilute mineral acid is used. Which Method is used in its collection? Give a reason.
(b)Explain the chemical preference of decorating a copper necklace with silver metal by using electrolysis method
(c)During electrolysis of brine, sodium was deposited at cathode and chlorine gas released at anode. If 2.0g of sodium were collected at cathode; find the volume of chlorine gas at s.t.p.
7. (a) Neutralization is applied in various useful situations with the aid of balanced chemical equation where necessary; describe any four usefulness of neutralization.
(b) 2.91 g of a monobasic acid, HX, were dissolved in water and made up to 250 cm3 with water. This solution was titrated with 0.108 M sodium hydroxide solution. 25 cm3 of the sodium hydroxide solution required 22.5 cm3 of the HX solution for complete neutralization. The equation for the reaction is.
HX (aq) + NaOH(aq) → NaX(aq) + H2O(l)
(a) Calculate the concentration in (i) g l-1 (ii) mol l-1 of the acid.
(b) Calculate the molar mass of HX.
8. (a) Gas A was prepared in the laboratory by isolating it from atmospheric air. During it's preparation air was allowed to pass through sodium hydroxide then over heated copper metal.
(i) Identify gas A.
(ii) By using equation, explain what happened when gas A passed through sodium hydroxide and in heated copper metal.
(iii) Write two uses of gas A
(b) (i) What will happen when copper is strongly heated in air at higher temperature and lower temperature respectively.
(ii) Explain how the pure copper is obtained from its ore?
SECTION C (30 MARKS)
Answer only two questions
9. (a) Explain the effects of the following to the environment:
- Acid rain
- Artificial fertilizers
- Plastic containers
(b) (i) What is the importance of ozone layer in the earth’s atmosphere?
(ii) Explain the effect of destroying the ozone layer.
(iii) Why does the increase of the concentration of carbon dioxide gas in the atmosphere results into increase of the earth’s surface temperature?
(iv) What control measures should be taken to prevent the destruction of ozone layer? Give three (3)points
(c) (i) Define: “ green house effect”
(ii) Give at least two (2) examples of gases which cause green house effect.
10. There are various environmental plans to be used in controlling the liquid and solid wastes in municipalities. Advise accordingly the Effective plans to be taken in order to achieve such activities. Give six points.
11. (a) Addition of inorganic fertilizers in the farm is not as important as addition of organic manure. Discuss the correctness of this statement in four(4) points
(b) Soil fertility and soil productivity are mistakenly used to mean the same concept. How do they differ from each other? Give five points
FORM FOUR CHEMISTRY EXAM SERIES 154
FORM FOUR CHEMISTRY EXAM SERIES 154
 | PRESIDENT’S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT MVOMERO DISTRICT COUNCIL |  |
FORM FOUR MOCK EXAMINATION
CODE: 032 CHEMISTRY
Time:3:00 Hours MAY. 2023
Instructions
- This paper consists of sections A, B and C with a total of eleven (14) questions.
- Answer all questions in section A and B and any One (1) question from section C.
- Section A carries Fifteen (15) marks, section B carries seventy (70)Marks and section C carries fifteen (15) marks.
- Non programmable calculators may be used.
- All communication devices and any unauthorized materials are not allowed in the examination room.
- Write your NAMES on every page of your answer booklet.
- The following constants may be used.
- Atomic masses:
Fe = 56, S = 32, O = 16, Na = 23, H = 1, Cl = 35.5, Mg = 24, Al = 27 P = 31 Pb = 207
- Standard pressure = 760mmHg
- Standard temperature = 273k
- 1 litre = 1 dm3 = 1000 cm3
SECTION A (15 Marks)
Answer all questions in this section
- For each of the items (i) – (x), choose the correct answer from among the given alternatives and write its letter besides the item number in the answer booklet provided.
- Which of the following is the correct sequence of the two steps you would follow during the scientific procedure?
- Hypothesis formulation and conclusion
- Observation and problem identification
- Experimentation and conclusion
- Problem identification and conclusion
- Which of the following sets of processes uses a gas that ignites with a “pop sound” when a lighted splint is passed through?
- Balloon filling, welding and diving
- Hardening oil, balloon filling and welding
- Hardening oil, balloon filling and diving
- Fueling rocket, diving and welding
- Balloon filling, fueling rocket and diving
- Oxygen gas can be produced at a large scale by
- Condensation of air
- Condensation of liquefied air
- Liquefaction of steam
- Fractional distillation of liquefied air
- Evaporation of liquefied air
- The basic causes of permanent hardness of water is:-
- Ca(OH)2
- Ca(HCO3)2
- Mg(HCO3)2
- Na2SO4
- CaSO4
- An element in the periodic table with atomic number 18 belongs to which of the following?
- Group I and period 1
- Group 0 and period 3
- Group III and period 3
- Group V and period 4
- Group VII and period 4
- Domestic utensil made up of iron rust easily as a result of the presence of:-
- Air and fire
- Air and water
- Water and Oil
- Air and Oil
- Water and oil.
- What is the correct meaning of ionization energy?
- Energy required to remove an electron from the inner most shell
- Energy required to remove electron from the outer most shell
- Energy required to add electron to the inner most shell
- Energy required to add electron to the outer most shell
- Energy required to attract electron towards the nucleus of an atom
- An increase in temperature of a gas in enclosed system/container causes an increase in pressure of the gas. This is because it increase the.
- Number of gas molecules
- Combination of gas molecules
- Number of collision between gas molecules
- Average velocity of a gas molecules
- Kinetic energy of the gas particles
- Which of the following is not the use of chromatography?
- To analyze blood in crime scenes
- To detect different fibres
- To detect water pollution
- To bleach dye or colour
- To test purify of organic substance
- Which of the following pairs constitute the best methods for treating and purifying water?
- Chlorination and aeration
- Chlorination and decantation
- Chlorination and filtration
- Chlorination and sedimentation
- Chlorination and distillation
2. Match the items in List A with responses in List B by writing the letter of the correct response beside the items number in the answer booklet provided.
| LIST A | LIST B |
|
|
SECTION B (70 Marks)
Answer all questions in this section
3. You are provided with the following fuels fire woods, natural gas, charcoal and kerosene.
- Giving three reasons, explain why natural gas is the best option among the fuels given above for domestic purpose.
- Giving four reasons, explain why fire wood and charcoal which are cheap and most available fuels are not recommended for domestic uses.
4. (a) Janeth heated compound “W” in a test tube and observed that a brown gas was produced, residue X formed. Also when she inserted a glowing splint into the test tube rekindled.
- Which gas rekindled the glowing wooden splint in the test tube.
- Identify compound “W” and residue “X”
- Write down a chemical equation for the reaction that took place.
- Explain the observations that would be made when aqueous ammonia is added drop by drop until in excess on a solution W
5. (a)Explain the concept of isomerism
(a)Draw the condensed and open structure for each of the following compounds
- 3-ethylpent-2-ene
- 4-methylhex-2-ene
- 5-methylhex-1-ene
6. (a) A student was preparing food for the family using hot oil on a frying pan. Accidently, the pan tipped over and a huge fire spread on the kitchen floor.
- Which fire extinguishers would be suitable for putting out the fire? Explain
- Which fire extinguishers would not be suitable for putting out the fire? Explain
(b) The following results were obtained in an experiment to find the molecular formula of magnesium oxide when magnesium burnt in air:-
Mass of crucible = 12. 6g
Mass of crucible + magnesium = 14.9g
Mass of crucible + magnesium oxide = 16.5g
- What mass of oxygen combined with magnesium?
- What was the empirical formula of this oxide?
7. (a) Different salts behave differently when heated. Use the balanced Chemical equation to show how carbonates and sulphate behave when Subjected to heat.
- Write a balanced and ionic equation for reaction between sodium carbonate and hydrochloric acid.
- Differentiate ionic equation from molecular equation.
8. (a) Why does a mixture of iron and sulphur after heating starting to glow and continues to glow even after the external source of heat is removed?
(b). Write the balanced equation for the reaction that takes place in (a) above.
(c). pure nitric (V) acid is colourless liquid. Explain why the one prepared in the laboratory is yellow?
9. Two experiments A and B were conducted to prepare hydrogen gas by varying the size of zinc granules which were reacted with dilute hydrochloric acid. All other factors were kept constant in the two experiments. Data obtained were used to plot the following graph.
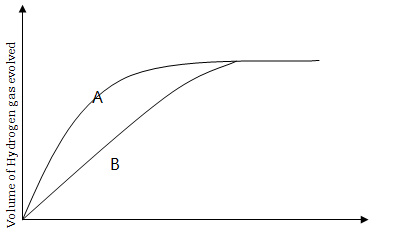
- Briefly explain the difference in the results of experiment A and B
- What factors can be adjusted to increase the yield of the product?
10. (a) Explain any five uses of chlorine gas
- Write an equation to show the reaction between chlorine gas and potassium bromide.
11. (a) Putin likes to use hard water to wash his clothes rather than soft water. His wife told him
that, hard water has higher cost than soft water. Give three reasons to support Putin’s wife.
(b) Farida went to the laboratory and found four bottles on the bench, each contain the mixture of the following: Bottle A contains Iodine and sand, Bottle B contains Alcohol and water, Bottle C contains iron fillings and powdered calcium carbonate and Bottle D contains Kerosene and water. Which methods can be used by Farida to separate the mixture of each bottle above?
12. (a) Makame’s daughter was sick. When he took her to the hospital, she was
Prescribed some medicine including a bottle of syrup. The bottle was written “shake well before use”.
(i) What does this statement signify?
(ii) Give two differences between the prescribed medicine in 10(a) above and the mixture of sugar and water.
(b) Students of mining course at a certain college conducted research on extraction of iron. They found that iron can be obtained from its ore in four stages. Briefly explain to them the four stages they should follow to extract it.
SECTION C (15 Marks)
Answer one (1) question from this section
13. A compound M is composed of 26.53% potassium, 35.37% chromium and the rest is Oxygen. If one fourth of its molecular mass (one second of its vapour density) is 73.5.Determine the molecular formula of compound M. solution of hydrated Sodium carbonate (Na2CO3.XH2O) by dissolving 38.9 g to make one litre of solution. 25 cm3 of hydrated Sodium carbonate solution was titrated with 0.24M hydrochloric acid by using POP indicator. The following results were obtained
| Volume Reading | Pilot | I | II | III |
| Final (Cm3) | 28.95 | 28.40 | 28.45 | 38.50 |
| Initial (Cm3) | 0.00 | 0.00 | 0.00 | 10.00 |
| Used (Cm3) | ||||
Questions
I. Find the average volume of acid
II. Write balanced equation for reaction
III. Calculate number of moles of acid used in the reaction
IV. Calculate number of moles of base used in the reaction
V. Calculate molarity of base (mol/dm3)
VI. Calculate concentration (g/dm3) of base
VII. Calculate relative molecular mass of the hydrated metal carbonate
VIII. Calculate relative atomic mass of X.
14.(a) Briefly explain the following observation about a sample of hard water.
- When boils, it forms white precipitates.
- After boiling , water forms a scum.
- Sodium carbonate makes the water completely soft.
(b).With the aid of chemical equations, briefly describe the following processes
- The removal of temporary hardness of water by boiling.
- The removal of permanent hardness of water by chemical means.
FORM FOUR CHEMISTRY EXAM SERIES 125
FORM FOUR CHEMISTRY EXAM SERIES 125
PRESIDENT'S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
KILIMANJARO REGION MOCK FORM FOUR EXAMINATION
CODE:032/1 CHEMISTRY 1
TIME:3:00HOURS MAY 2023
INSTRUCTIONS
- This paper consist of sections A, B,and C with a total of eleven(11) questions.
- AnswerALL questions in section A and B and any TWO (2) questions from section C.
- Section A carries sixteen (16)marks, section B carries fifty four (54) marks and section C carries thirty (30) marks.
- Non programmable calculators may be used.
- Cellular phones and any unauthorized materials are not allowed in the examination room.
- Write your examination number on every page of your answer sheet(s).
- The following constants may be used.
- Atmicmasses:H=1, C=12, N=14, 0=16, Na=23, S=32.
- GMV ats.t.p=22.4dm3.
- 1 Faraday=96,500 coulombs
- 1 litre =1 dm3=1000 cm3.
SECTION A (16 Marks)
Answer ALL questions in this section
1. 1.For each of the items (i) - (x) choose the correct answer from thegiven alternatives and write its letter beside the item number in the answer sheet(s) provided
(i) When water is added to an acid, the acid becomes
- More acidic and its pH goes down
- More acidic and its pH goes up
- Less acidic and its pH goes down
- Less acidic and its pH goes up
- Neutral and its pH become 7
(ii) 1.4g of potassium hydroxide is dissolved in water to form 250cm3 of Solution.
What is the Molarity of this solution?
- 0.001M
- 0.1M
- 1.4M
- 5.6M
- 6.0M
(iii) An electric current was passed through a concentrated solution of hydrochloric acid using carbon electrodes. The substance liberated at anode was.
- Copper
- Hydrogen
- Oxygen
- Sodium
- Chlorine
(iv) If element Q of group (H) combines with element R of group (IV) what will be the formula of the resulting compound.
- R2Q
- QR6
- R3Q
- R3Q
- Q2R
(v) The process of giving away water of crystallization to the atmosphere by a chemical substance is called.
- Efflorescence
- Deliquescence
- Hygroscope
- Sublimation
- E.Vaporization
(vi) Which of the following elements does not react with chlorine?
- Aluminum
- Oxygen
- Sodium
- Argon
- Iron
(vii) When methane undergoes substitutional reaction with excess chlorine „what is the final product?
- Chloromethane
- Dichloromethane
- Trichloromethane
- Tetrachloromethane
- Monochloromethane
(viii) Scum tends to be formed in washing clothes with hard water due to the reaction between
- Hard water with soap
- Calcium ions, magnesium ions and sodium stearate
- The dissolved calcium and magnesium salts with detergents
- Hydrogen carbonate ions and soap
- Hydrogen carbonate ions and clothes
(ix) The IUPAC name of H2SO4 is:
- Sulphuric (IV) acid
- Sulphuric acid
- Sulphuric (V) acid
- Sulphorous acid
- Sulphuric (VI) acid
(x) Which of the following are the components needed to start fire?
- Match box, fire wood and kerosene
- Match box, fire wood and oxygen
- Oxygen, fuel and heat
- Oxygen, fuel and fire wood
- Heat, match box and oxygen
2. Match the items in List A with their responses in List B by writing the letter of the correct response beside the item number in the provided answer sheet(s)
| LIST A | LIST B |
|
|
SECTION B (54 Marks)
Answer ALL questions in this section.
3. (a)The headmaster of Mwembeni secondary school is planning to build a Chemistry laboratory.You as a young chemist advise him on how the Laboratory features like window and doors are supposed to be. Give Reasons for your advice.
(b) Mussa placed a large crystals of potassium permanganate in the Bottom of beaker of cold water and left it for several hours.
- What he must observed after five minutes? Explain.
- What he must have observed after several hours? Explain.
- Name two processes which have taken place during experiment.
4. (a)People who use hard water can expect higher costs than people who Use soft water. Briefly explain by giving three reasons.
(b) 25cm3 of water sample A, B, C and D were tested with soap solution.The volume of soap solution required to produce lather that lasted for minutes was recorded.Fresh sample of each were boiled and tested again with soap solution.The results are shown in the table below:
| Sample | Observation with soap Solution | Observation for boiled water + soap Solution |
| A | 5.0 | 5.0 |
| B | 1.0 | 1.0 |
| C | 11.0 | 8.0 |
| D | 9.0 | 1.0 |
- Which sample probably contains temporary hardness of water only?
- Which sample probably contains both permanent and temporary hardness of water? Give a reason for your answer.
5. (a)Sodium chloride (NaCI) and hydrogen Chloride (HCI) are both chloride Compounds.Differentiate the two compounds by giving three reasons.
(b)(i)Differentiate empirical formula from molecular formula. cetain compound formed by Sulphur and Oxygen contains 40.1% sulphur by mass.Workout the empirical formula of this compound.
6. (a)(i)Organic chemistry is very essential in both industrial and life processes. Give reasons by using three points.
(ii)Why carbon has been given special attention in organic chemistry rather other elements. Give four reasons.
(b) Given .organic compound with molecular formula C3H7OH. Give its IUPAC Name,functional group and its general formula.
7. (a)(i)Differentiate endothermic from exothermic reaction.
(ii)State Le-Chatelier's principle.
(b)Consider the following reversible reaction:
![]()
Will the production of NH3 be affected if Pressure changed?.
8. a)In practice , not all ions arrive at a particular electrode are discharged. Give three reasons which may decide the ion .to be preferentially Selected for discharge.
b). In the electrolytic refining of copper, anode was made of impure copper of 2g while cathode was made of pure copper of 1.5g. At the end of the experiment cathode was found to weigh 3g. Calculate the percentage purity and impurity of the impure copper.
SECTION C (30 Marks)
Answer only TWO (2) questions in this section
9. a)Metals are extracted. from the sea and in earth.Referring to Tanzania as among the countries in the world extracting metals, explain four stages of extraction of metals.
b). Does the extraction of gold follow all four stages? Give reasons.
10. Villagers want to apply fertilizer to their farms .As a chemist explain how this could be done.
11. Water in the lake near your village is not safe for human consumption and domestic activities. In five points explain what make water unsafe?
FORM FOUR CHEMISTRY EXAM SERIES 118
FORM FOUR CHEMISTRY EXAM SERIES 118
PRESIDENTS OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT

COAST REGION
FORM FOUR SECONDARY EDUCATION MOCK EXAMINATION
Code: 032/1 CHEMISTRY
(For Both School and Private candidates)
Time: 3 Hours
INSTRUCTIONS
- This paper consists of 11 (eleven) questions in sections A,B and C
- Answer ALL questions in section A and B, and only two (2) questions in section C
- Non-programmable calculators may be used in the examination room
- Cellular phones and any unauthorized materials are not allowed in the examination room
- Write your examination number on every page of your answer booklets
- The following constants may be used;
Atomic masses:
- H = 1, C =12, 16, S = 32, K = 39, Na = 23
- GMV at S.T.P = 22.4dm 3/mol
- 1 Litre = Idm 3 =1000cm 3
- 1 Faraday = 96500 Coulombs
SECTION A (15 Marks)
Answer ALL questions in this section
1. For each of the items (i) — (x), choose the correct answer from among the given alternatives and write its letter besides the item number in the answer booklet provided
(i) A beaker containing solid carbon dioxide is placed in a fume cupboard at room temperature. The carbon dioxide becomes gaseous. Which process describes this change of states?
- Boiling
- Evaporation
- Condensation
- Sublimation
(ii) Hamisi went to the laboratory for measuring specific volume of liquid X. What apparatus would be used to measure that liquid?
- Burette
- Volumetric flask
- Measuring cylinder
- Pipette
(iii) An aqueous solution turns red litmus solution blue. Excess addition of which of the following solution would reverse the changes?
- Vinegar
- Baking powder
- Aqueous arnmonia
- Lime
(iv) A simple proof that some chemical reactions take place in our bodies is that;
- We eat a balanced diet
- Waste products from our bodies
- We occasionally fall sick
- Doctors tell us so in the hospitals
(v) At home 20g of a table salt was mixed with 0.2 litre of water in a cup to form . . . . . . . . . . . . . .
- Suspension
- Heterogeneous solution
- Homogeneous solution
- Emulsion
(vi) The oxidation state of Nitrogen in the radical NH4+ is;
- "1
- +2
- +3
- "3
(viii) Group II elements burn in oxygen to form . . . . . . . .
- Metal oxides
- Non-metal oxides
- Hydroxide
- Carbon dioxide
(ix) Elements lose or gain electrons to form
- Isotopes
- Radicals
- Molecules
- Ions
(x) What name is given to the force of attraction that holds atoms together to form a compound?
- Chemical change
- Physical change
- Chemical bond
- Centripetal force
(xi) Class B fire can be extinguished by using each of the following;
- Carbon dioxide and sand
- Carbon dioxide and water
- Sand and water
- Oxygen and carbon dioxide
2. Match the items in LIST A with the responses in LIST B by writing the letter of the correct response besides the item number in the answer sheet provided
| LIST A | LIST B |
| (i) Biogas (ii) Biomass (iii) Natural gas producer gas (iv) Water gas (v) Ethanol |
|
SECTION B (54 Marks)
Answer ALL questions in this section
3. Aisha drew a periodic table and then put a shadow on the element with atomic number 8
(a) What type of chemical bond is found between atoms of the element?
(b) Compound X contains 24.24% Carbon, 4.04% Hydrogen and 71.72% Chlorine.
Given that, the vapour density of X is 49.5.
(i) Calculate molecular formula of the compound X
(ii) Draw and name the open structural formula of the possible isomers of compound X
4. One of the laboratory rooms at Utete Secondary school, having only the following features; slippery floor, no water supply and having small windows. Does the room qualify to be used as a chemistry laboratory? Give four points.
5. A chemistry student prepared an experiment to produce a gas by decomposing a compound using electricity. A steady current was allowed to flow through the solution for 3 hours. At S.T.P 4.12cm 3 of the gas which relights a glowing splint was produced.
(a) (i) What terminology is used to refer such an experiment set up?
(ii) Work out the current flowing through the circuit
(b) Classify the following salts in terms of their solubility; Silver chloride, Lead nitrate, Zinc chloride and Barium sulphate
6. (a) Despite its expensiveness, hard water treatment is very important in large scale. Explain three (3) importance of hard water treatment in a day to day life.
(b) What are the three (3) methods used for softening permanent hard water?
(c) With the aid of chemical equation, show any method for softening permanent hard water
7. (a) Iron is extracted from various ores by reduction in the blast furnace.
- What is the chief ore from which iron is extracted?
- Write the equation for the reduction of the ore in the blast furnace
- Explain the role of the following substances in the blast furnace; Limestone and coke.
(b) If 0.5g of hydrogen gas is exposed in air, what mass of water will be formed?
8. (a) An unkown white powdered sample was mixed with dilute nitric acid (HN03) and gave a colourless solution and a gas which precipitated lime water.
- Identify the white powdered sample and gas evolved
- Give a balanced chemical equation for the reaction above
(b) Why is the use of the balanced chemical equations so important in chemical manufacturing industries? Give four (4) reasons.
SECTION C (30 Marks)
Answer any TWO (2) questions from this section
9. How can you tell that water is polluted? Give two (2) ways.
(b) We have coal at Kiwira in Mbeya region. Government authorities have allowed use of coal for domestic and industrial purpose. What warning can you raise concerning the likely effects? Give five (5) points.
10. Suppose the pond water in your area is contaminated and has caused diseases to the people. How would you solve this problem using scientific methods?
11. (a) Why Carbon has been given special attention in organic chemistry rather than other elements?
(b) You are provided with the following compounds, A, B, C and D which correspond to the molecular formula C2H6, C2H4, C2H5OH and CH3COOH respectively.
- Give the IUPAC names of compound C and D. Write the chemical equations for the reaction between
- C and concentrated H2SO4
- B and hydrogen gas in presence of Nickel at 200 0C
- C and D at 1800C
- C combusted at limited amount of air
(c) What kind of reaction has taken place at b(ii) and b(iii) above?
FORM FOUR CHEMISTRY EXAM SERIES 111
FORM FOUR CHEMISTRY EXAM SERIES 111
THE UNITED REPUBLIC OF TANZANIA PRESIDENT'S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
TANGA REGION

FORM FOUR MOCK EXAMINATION 032/1 CHEMISTRY I
(For Both School and Private Candidates)
TIME: 3 IIOURS MAY 2023
INSTRUCTIONS
- This paper consists of section A, B and.0 with a total of eleven (11) question
- Answer All questions in section A and B and two (2) questions from section C
- Section A Carries sixteen (16) marks, B (54) marks and section C (30) marks
- Write your examination number at the top Corner of every page of your answer sheet
- Cellular phones and Smart watch are not allowed in the examinations rooms, non programmable calculators may be used.
- You may use the following
- Atomic masses C = 12, H = 1, N = 14, Cl = 35.5, Na = 23, 0 =. 16, Zn = 65, Ca = 40, Cu = 64, S = 32, Ag = 108
- Avogadro's number = 6.02 x IO23
- GMV at S.T.P = 22.4dm3
- 1 Faraday = 96,500 Coulombs
- Standard pressure = 760 mmHg
- Standard temperature = 273K
- 1 Litre = 1 dm3 = 1000cm3
SECTION A (16 MARKS)
Answer all Questions in this section
1. For each item (i-x), choose the correct answer from the given alternatives and write the letter beside the item number in the answer booklet.
i. The solution with pH of 5 is said to be:,
- A strong base
- A neutral
- A weak acid
- A strong acid
- A weak base
ii. Ethanol react with Ethanoic acids to form a group of organic compounds called
- Alkynes
- Halo alkanes
- Esters
- Alkenes
- Alkanes
iii. Which of the following is NOT a component of the first aid kit?
- Goggles
- A pair of scissors
- Dropper
- Gloves
- Razor blade
iv. Which among of the following chemical reactions rapidly releases energy in form of light and heat?
- Combustion
- Decomposition
- Displacement
- Neutralization
- Precipitation
v. Laboratory Technician prepared a solution containing 26.5g of anhydrous Sodium carbonate in 5 dm3 of the solution and provided to Form Four students to calculate Its Molarity. Which among the following will be the possible answer?
- 0.05
- 0.25
- 1.25
- 5.3
- 0.025
vi. Why oxygen differs from other gases?
- It neither burns no support combustion
- It supports combustion but does not burn
- It burns but does not support combustion
- It burns and support combustion
- It exploded and support combustion
vii. The oxidation state of Chlorine in Sodium chlorate (NaCIO3)is:
- -1
- +2
- +5
- +3
- -3
viii. Elements loss or gain electrons to form:
- Isotopes
- Radicals
- Molecules
- Ions
- Allotropes
ix. Insoluble salts like Barium sulphate, generally can be obtained in the laboratory by:
- Evaporation of its concentrated solution
- Crystallization
- Precipitation
- Decomposition
- Displacement reaction
x. In a blast furnace Carbon monoxide is prepared by passing Carbon dioxide over a red hot coke. What is the chemical role of Carbon dioxide?
- An accelerator
- An oxidizing agent
- A reducing agent
- A catalyst
- Oxidized
2. Match the items in list A with responses in list B by writing the letter of the correct response beside the item number in the answer booklet provided
| LIST A | LIST B |
| (i) N2(g) + 3H2(g) → 2NH3(g) | A. Esterification reaction |
| (ii). 2Pb(NO3)2(s) | B. Displacement reaction |
| (iii) AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq) | C. Precipitation reaction |
| (iv) Fe(s) → F3-(aq) +3e- | D. Decomposition reaction |
| (v) Zn(s) +2HCl(aq) → ZnCl2(aq) + H2(g) | E Combination reaction |
(vi) CH3COOH(aq) ? CH3COOCH3(aq) + H2O(l) | F. Redox reaction |
SECTION B (54 MARKS)
Answer all questions in this section, each question carry 9 marks
3. (a) Write ionic equation for the precipitation of Barium sulphate from Barium chloride and Sodium sulphate
(b) It is not advisable to sleep inside the house which is not well ventilated with a burning wooden charcoal. Give a reason for that and write the chemical equation to support your answer.
(c) Consider the following elements of group seven n order of which they appear in their group in the periodic table F, C 1, Br, and I
- Which element is more electronegative
- Name the least electronegative element
- Which element has the largest atom?
- Write the electronic configuration of the chlorine atom (9marks)
4. (a) Distinguish between Temporary Hardness and Permanent Hardness of Water basing on their ions.
(b) By use of equations, show how each of the type of hardness in (a) above can be eliminated.
(c). Giving four reasons, explain why people who use hard water can expect high costs than people who use soft water. (9 marks)
5. (a)Which homologous series of organic compounds can be represented by the following general formula?
- CnH2n+2
- CnH2n
- CnH2n+1OH (3 marks)
(h) Give the name of the first compound in each series. (3 marks)
(c) (i) Describe a reaction by which a named compound of series in (a) (ii) can be converted to a compound of series in (a) (i).
(ii) How can a compound of series (a)
(iii) he converted to a compound of series in (ii)? (3marks)
6. (a) You are provided with a compound composed of 22.2% Zinc. 11.6% Sulphur, 22.3% oxygen and the rest percentage is water of crystallization. Calculate the molecular formula of the compound if its Molecular mass is 283.
(b) Why it is important to balance the chemical equation'? Provide three points to support your answer.
(c) If 0.5g of Hydrogen gas is exploded in air. what is the mass of water formed (9 marks)
7. (a) Ammonia gas is manufactured by Nitrogen gas with Hydrogen gas in the presence of a catalyst. Write a balanced chemical equation for the reaction and explain the. role played by the catalyst in this reaction.
(b) When Hydrogen Sulphide gas is passed through Sulphur dioxide a yellow deposit of Sulphur is produced immediately. Write the chemical equation for this observation
(c) Give the name of the metal which meets the following requirements
- The metal which must be kept in kerosene to protect it from air and water
- The metal which is found in limestone
- The metal of which its oxide is brown when hot and yellow when cold (9 marks)
8. (a) Element X has 20 electrons and mass number of 40. Work out the number of each type of nucleons present and identify whether element X is a metal or.non-metal
(b) In the industrial preparation of Sulphur trioxide Mr Atieno etablished equilibrium between Sulphur dioxide and oxygen gas as follows:
2SO2(g) + O2(g) ? 2SO3(g) ?H = -94.9KJ/mol
- Explain the way of maximizing the proportion of the product at equilibrium?
- Why is it unfavorable to work with very low pressure and very high temperature in the contact process?
- What is the catalyst used to speed up the rate of formation of Sulphur trioxide before attaining the equilibrium? (9Marks)
SECTION C (30 Marks)
Answer two (2) questions from this section
9. Soil Genesis deals with factors and processes of soil formation. The formation of soil is the result of the Interaction of several factors. Explain five (5) factors for soil formation.
10. We have Coal at Kiwira in Mbeya Region. Authorities in the Government have allowed use of coal for domestic and industrial purpose. What warning can you raise concerning likely effects? Give five Points
10. (a). When I was in an interview at a certain school, one student asked me to State Faraday's Laws of Electrolysis. If you were me how could you answer that student?.
(b) Dilute Silver Nitrate solution was decomposed by the passage of electric current through it. What mass of Silver and what volume of Oxygen (Measured at S.T.P).would be liberated in electrolysis by 9650 Coulombs of Electricity?
(c). Mr. Chonya said that Electrolysis is applied in many fields in our daily life. Provide four evidences to support this statement.
FORM FOUR CHEMISTRY EXAM SERIES 93
FORM FOUR CHEMISTRY EXAM SERIES 93
THE UNITED REPUBLIC OF TANZANIA PRESIDENT'S OFFICE, REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
ZONAL FORM FOUR MOCK EXAMINATION
(KATAVI, MBEYA, NJOMBE, RUKWA AND SONGWE REGIONS)
CHEMISTRY
Time 3:00 Hours Year: 2022
INSTRUCTIONS
- This paper consists of Section A, B and C with a total of fourteen (14) questions
- Answer all questions in Section A and B and One (1) question from Section C.
- Cellular phones and any unauthorized materials are not allowed in the examination room.
- Write your Examination Number on every page of your answer booklet (s0
- The following constants may be used.
- Atomic masses: H=1, O=16, N=14, S=32, Zn=65, Cl=35.5, Cu=63.5, Ag=108
- Avogadro’s number = 6.02 x 1023
- GMV at s.t.p. = 22.4dm3
- 1 Faraday = 96,500 coulombs
- 1 Litre = 1dm3 = 1000 cm3
SECTION A: (15 MARKS)
Answer all question in this section.
1. For each of the items (i) – (x), choose the correct answer from the alternatives and write its letter beside the item in the answer sheet provided.
(i) The best chemical warning signs that should be put on bottles containing kerosene is ……….
- Corrosive
- Toxic
- Flammable
- Explosive
- Harmful
(ii) A juice from Bakhresa Company written “shake well before use”. What does it mean?
- Suspension
- Solution
- Solute
- Emulsion
- Solvent
(iii) Alcohol reacts with carboxylic acid to form a group of organic compounds called
- Alkynes
- Halo alkane
- Esters
- Alkane
- Ethers
(iv) Ammonia gas is collected by which method among the following?
- Downwards displacement of water
- Upward delivery
- Downward delivery
- Upward displacement of air
- Collected over water
(v) Microelements from the soil are required by plants for their proper growth and development. The term microelements means ………
- The elements are very small
- The elements are required in small amount
- The elements are very rarely found in the soil
- The elements are not required by adult plants
- The elements are required by adult plants.
(vi) What is the oxidation number of the phosphorus in the following compound H3PO4?
- -5
- 0
- +2
- +5
- -3
(vii) What volume of hydrogen gas will be produced when 1.3g of zinc granules react completely with excess dilute sulphuric acid at s.t.p?
- 223cm3
- 130cm3
- 220cm3
- 440cm3
- 448cm3
(viii) Naima was preparing some food for her family using cooking oil in a frying pan. Accidentally the tipped over and a huge fire spread over her kitchen floor. Which extinguishers should be appropriate for putting off the fire?
- Wet sand carbon dioxide
- Dry powder and carbon dioxide
- Sand and wet chemicals
- We sand dry powder.
(ix) Which of the following is not organic compound?
- CO
- C6H12O6
- CH4
- CH3COOH
- C2H5Br
(x) The following reaction 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O (l) is an example of a
- Redox reaction
- Combination reaction
- Esterification
- Neutralization reaction
- Decomposition reaction.
2. Match the item with List A with responses in List B by writing the letter of the correct response beside the corresponding item number in the answer sheet provided.
| LIST A | LIST B |
|
|
SECTION B: (70 MARKS)
Answer all questions in this section
3. (a)Catherine is planning to make fire for cooking ugali for her family. What are necessary conditions which must be present so that she can make fire successfully for cooking ugali for her family?
(b)Give for each of the followings
- Water is universal solvent
- Some metal like zinc do not get rust.
- Chlorine gas is collected by downward delivery
- Carbon dioxide turns lime water into milky colour.
4. (a) 25cm3 of 0.1MHCl were neutralized by 23cm3 of Na2CO3 solution. Calculate the concentration of the alkali in grams per litre.
(b)Suggest a suitable indicator of each of the following titrations
- Hydrochloric acid against ammonia solution
- Sulphuric acid against sodium hydroxide solution
- Ethanedioic acid against potassium hydroxide solution
5. (a)State the modern periodic law
(b)Study the periodic table below then answer the questions that follow.
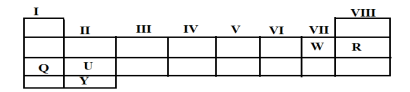 |
Write the formula of compounds formed when element
- U and W combine together
- Q and W combine together
Draw and write electronic configuration of Y.
6. (a)What do you understand the term chemical equation?
(b)Write ionic equation for the following chemical reactions.
- Milk of magnesia is used to relieve indigestion
- A divalent metal displacing monovalent metal from its soluble nitrate
7. (a)Define the following
- Soil fertility
- Leaching
(b)Briefly explain how soil PH is important? (Give three points)
(c)List four methods of fertilizer application.
8. The table below shows brads of bottled water for drinking and the concentration of different minerals ions in each brand. Study the table and then answer questions below it.
| Composition In mg/litre | Mineral | Na+ | Ca2+ | Mg2+ | Cl- | NO3- | SO42- | Fe2+ | F- |
| UHAI | 40.0 | 3.05 | 4.15 | 14.18 | 0.48 | 10.0 | 0 | 1.76 | |
| DASANI | 22.32 | 2.69 | 0.11 | 6.50 | 1.0 | 8.0 | 0 | 0.45 |
9. (a)Give good reason (s) for the following. (Answer according to the question demand). Natural gas is so popular in heating and cooking in homes. (2 points)
- Natural gas is so popular in heating and cooking in homes. (2 points)
- Nuclear energy is not a sustainable source of energy. (2 points)
- Coal and petroleum are non-renewable sources. (1 point)
(b) State the main raw materials and process involved in the manufacture of each of the following products.
- Wood charcoal
- Coke
- Lamp black
- Animal charcoal
10. (a) Explain any three (3) economic importance of electrolysis.
(b)A current of 0.5A were made to flow through a silver voltammeter for 30 minutes. Calculate the mass of silver deposited at cathode.
11. (a)An organic compound is represented by the general formula CnH2n+2 (in each case n=1, 2, 3 ….). Write down the condensed structure formula and IUPAC names of the isomers of the fourth member from the above general formula.
(b)Write down the structures of the following compounds
- 5-iodo-4-methylhex-l-yne
- 6-methyl-4-propyloct -2-ene
- 2-fluoro-2, 3, 5, 5- tetramethylhexane.
12. During a practical session a tea spoon of common salt was being dissolved in 100mls of water but at different temperature to determine the effect of temperature on the solubility of a salt as shown below.
| Volume of water (mls) | Temperature of water (°C) | Solubility of salt |
| 100 | 20 | 5 |
| 100 | 30 | 10 |
| 100 | 40 | 15 |
| 100 | 50 | 20 |
| 100 | 60 | 25 |
- Classify volume of water, temperature and solubility of salt into; dependent, independent and controlled variable
- Explain the application of scientific procedure in everyday life. (4 points)
SECTION C: (15 MARKS)
Answer only one question from this section.
13. What are the causes, effects and remedial measures of water pollution? (Give at least two points from each category)
14. (a)Explain the meaning of each of the following terms.
- Reversible reaction.
- Dynamic equilibrium
(b)The industrial oxidation of Sulphur dioxide is summarized in the equation below.
2SO3(g) + O2(g) ![]() 2SO3(g) H=-94.9 Kjmol-1
2SO3(g) H=-94.9 Kjmol-1
What will be the effect of each of the following on the production of Sulphur trioxide?
- Increase in moles of Sulphur dioxide
- Increase in pressure
- Decrease of temperature
- Decrease of moles of Sulphur dioxide.
(c)Briefly explain how each of the following factors affects the rate of a chemical reaction.
- Temperature
- Pressure
- Concentration
- Catalyst
- Surface area
(d)Give one good reason for the following
- Fruits ripe faster during summer than during winter
- Steel wire get rust faster than iron nails.
FORM FOUR CHEMISTRY EXAM SERIES 79
FORM FOUR CHEMISTRY EXAM SERIES 79
PRESIDENT'S OFFICE, REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
ARUSHA REGIONAL FORM FOUR MOCK EXAMINATION
032 CHEMISTRY 1
TIME : 3 Hours July 2022
INSTRUCTIONS
- This paper consists of section A, B and C with a total of fourteen (14) questions.
- Answer all questions in section A and B and one question from section C.
- Section A carries 15 Marks, section B carries 70 Marks and section C carries 15 Marks.
- All writing must be in black or blue ink except diagrams which must be in pencil.
- Cellular phones and any unauthorized materials are not allowed in the examination room.
- Non — Programmable calculator may be used.
- Write your NAME on every page of your answer sheets.
- The following constants May be used.
Atomic masses.
- 0=16, H----1,C1-35.5, S-32, C-12, N = 14,Ca = 40,
- Na=23, Cu = 64, Zn —65, S = 32,K= 39
- Avogadro's constant = 6.02 x 1023 G.M.V. at S.T.P. = 22.4 dm3
- 1 Faraday = 96500 coulombs 1 Litre ---- 1 dm3 = 1000 cm3
SECTION A (15 Marks)
Answer ALL questions in this section
For each of the items (i) — (x), choose the correct answer among the given alternatives and write its letter beside the item number in the answer sheet provided.
(i) Oxygen gas can be produced at a large by;
- Condensation of air
- Condensation of liquefied air air
- Liquefaction of steam
- Fractional distillation of liquefied
- Evaporation
(ii) Which of the following substance should not kept closely to the open bottle containing carbon-dioxide;
- Dilute nitric acid
- Dilute hydrochloric acid
- Sodium hydroxide solution
- Sodium nitrate
- Dilute sulphuric acid
(iii) The substance that can burn your skin is best described as: -
- Corrosive
- Explosive
- Flammable
- Toxic
- Oxidant
(iv) 10cm3 of 0.4M Sodium Hydroxide are added to 40cm3of 0.2M hydrochloric acid. The resulting mixture will be
- Neutral
- Alkaline
- Dilute
- Acidic
- Amphoteric
(v) Which of the following Chemical species have the same number of electrons?
- K+, Ca2+, Cl- and Ar
- O2-, Ca2+, Cl- and Mg2+
- Na+, Mg2+, Be2+and Na
- Cl, Na, K+and A13+
- K+, Ca2+, CI and Ne
(vi) When burette is used in the laboratory its calibration starts from above because.
- It measures the volume of remained
- It measures the used volume
- It measures the volume added
- It measures the volume of base added
(vii) Consider the following fuels which are used for-different purposes
1, Charcoal 2, Diesel 3. Coal 4. Fire wood
Which fuels are considered as secondary fuel?
- 1 and 4
- 1 and 3
- 2 and 3
- 3 and 4
- 1 and 2
(viii) A metal nitrate which will not give a metal oxide on heating is:
- Calcium nitrate
- Silver nitrate
- Lead nitrate
- Copper nitrate
- Zinc nitrate
(ix) When nitrogen gas is formed covalently how many electrons are shared between nitroge atoms.
- 2
- 3
- 6
- 5
- 4
(x) Which substance can be reduced when heated with carbon;
- Alminium
- Calcium carbonate
- Iron III oxide
- Magnesium oxide
- Sodium oxide
2. Match the items in List A with a correct response in List B by writing the letter of the correct response below the corresponding item number.
| List A | List B |
| (i) Class A (ii) Class B (iii)Class C (iv)Class D (v) Class E |
|
SECTION B (70 Marks)
Answer ALL questions in this section
3. (a) (i) What types of a chemical bond is found between fluorine atoms in a fluorine molecule?
(ii) Name other type of chemical bond formed by Fluorine with other elements. Give an example.
(b) Compound X contains 24.24% Carbon, 4.04% hydrogen and 71. 72% Chlorine. Give that the vapour density of X is 49.5
(i) Calculate the molecular formula of compound X
(ii) Name the molecular formula of compound X
(iii)Give the condensed structural formula of compound X
4. (a) Differentiate the temporary hardness of water from permanent hardness of water basing on their content.
(b) Consider the sample of water taken indifferent places at Arusha region. The samples were boiled and treated with the soap while others were treated with soap before boiling to obtain different results:
| Sample Of Water | Unboiled | Boiled |
Arusha Dc Oldonyo Sambu Maroroni Njiro | Volume of Soap used(cm3) 1.0 8.5 10.5 5.9 | Volume of Soap used(cm3) 1.0 8.5 3.8 1.0 |
(i) Which sample contains temporary hardness of water? (Give reason)
(ii) Which sample contains permanent hardness of water? (Give reason)
(iii) What do you think will be the cause of variation of volume of soap of Maroroni water?
(c) Mention the methods (s) used to soften sample of water at Njiro.
5. (a) Explain how you can differentiate base (alkali) from acid in the Laboratory using Methyl orange (MO) and phenolphthalein (POP) indicators.
(b) 25cm3 of impure Sulphuric acid containing 5.2g/dm3 reacted with 25cm3 of sodium hydroxide solution made by dissolving 2g of sodium hydroxide in 500cm3 of water Calculate percentage purity and impurity of the acid the acid.
6.(a) A student accidentally broke a beaker containing Copper II Sulphate crystals. He decided to separate the blue crystals from small pieces of glass by first dissolving the mixture. What were his next steps?
(b) In the industrial preparation of sulphur trioxide equilibrium is established between sulphur dioxide and oxigen as follows:
(i) Is the forward reaction endothermic or exothermic? Give reason
(ii)How could you adjust temperature and pressure to maximize the proportion of product at equilibrium
(iii)Why it is unfavorable to work with very high pressure and very low temperature in the contact process
(iv) What catalyst is used to speed up the rate of the formation of Sulphur trioxide.
7. (a) With example in each differentiate alloy from allotropes.
(b) Carbon is one of the elements that have allotropes. Explain how two main allotrope of carbon differ from each other (4 points)
8. (a) Name three gases which should be not produced in order to prevent the destruction of ozone layer. (b) Explain for effects of ozone layer destruction.
9. Study the information in the table below and answer the questions that follow.
| Element | Atomic number |
| V | 12 |
| W | 13 |
| P | 16 |
| R | 18 |
| 1 | 19 |
| M | 9 |
(a) State the elements which belongs to the same period
(b) (i) Identify element which is gaseous at room temperature
(ii) State an element which does not form an oxide
(iii) Give the chemical formula for the carbonate of element W.
(iv) Give a balanced chemical equation between the element which belong to alkaline earth metal and halogen above.
(c) Write balanced chemical equation when element I reacts with cold water.
10. (a) Consider the diagram below;
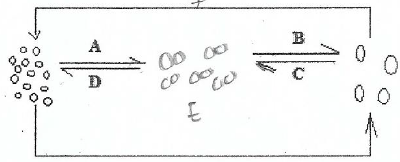
(i) Give aim of the above process
(ii) Identify the process A to F
(iii) Give two importance of the above diagram to our daily life.
(b) State whether the following is permanent change or temporary change,
(i) Dissolution of salt in water
(ii) Rotting of mangoes
11. The diagram shows a set up to demonstrate the reaction between Chlorine and hydrogen sulphide gas.
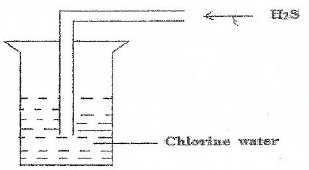
(a) What will be observed when hydrogen sulphide gas is bubbled through chlorine water.
(b) Write an equation for the reaction between Chlorine and hydrogen sulphide.
(c) Give the name of the solid product formed in (b) above and write two uses of it.
(d) Which substance is an oxidizing agent and reducing agent in the above reaction.
12. Dilute nitric acid is added to a green solid P. A blue solution R is formed and gas I precipitate with lime water is formed. The blue solution R is evaporated to dryness and in the Pyrex test tube to give black solid M, brown fumes of gas W and colourless gas glowing splint was formed.
(a) Identify the substance P, R, I, M, Wand S
(b) Write the chemical equation for the;
(i) Dilute nitric acid and solid P
(ii) Formation of white precipitate with gas I and lime water.
SECTION C (15 Marks)
Answer ONE (1) question from this section
13. With the aid of illustration, describe six methods of separating mixtures
14. (a) Give three (3) unique properties ca Carbon as a primary element in any organic compou, (b)Give the IUPAC name of the following;
(i) CH3C113 (iii)CH3COOCH3
(ii) CH3CH CH2 (iv)CC14
(c) Write the open structure formula of the following;
(i) Pent-1 -ene
(ii) 2, 2- dimethyl butan-l-ol
(iii)3 - methyl but -1-yne
(iv)2,3 - dimethyl hex - 2 - ene
(d) Complete and balance the following organic reaction.
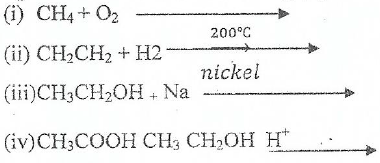
FORM FOUR CHEMISTRY EXAM SERIES 75
FORM FOUR CHEMISTRY EXAM SERIES 75
PRESIDENT'S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
DAR ES SALAAM REGION & IRINGA- REGIONS FORM FOUR JOINT MOCK EXAMINATION - 2022
CHEMISTRY - 1
CODE: 032/1
TIME: 3: 00 HOURS Friday 8th July 2022 A.M
INSTRUCTIONS
- This paper consist of sections A, B and C with a total of fourteen (14) questions
- Answer all questions in section A and B and one (1) question from section C
- Sections A and C carries Fifteen (15) marks each and section III carries seventy (70) marks
- Write your examination number of every page of your answer booklets
The following constants may be used.
- Atomic masses : [Na = 23, C = 12, 0 = 16, H = 1, CI = 35.5, Ag = 108, Cu = 64, Fe = 56, N = 14]
- Avogadro's number = 6.02 x 1023
- GMV at STP = 22.4 dm3
- 1 Faraday = 96500 Coulombs
- Standard pressure = 760 mmHg
- Standard temperature = 273K
- 1 litre = 1dm3 = 1000cm3
SECTION A (15 Marks)
Answer all questions in this section.
1. For each of the item (i) – (x) choose the correct answer from among the given alternatives and write its letter beside the item number in the answer booklet provided.
(i)Which of the following contains a list of elements with the same number of electrons
- CL, He, Ne, O2-
- Na+,Mg2+,Be2+,Li
- K+,Ca2+,Cl-,Ar
- O2-, F-, Ne, Na
- Cl2, Mg2+,Al, Na+, Li
(ii) The molarity of 5.3g in 100ml of Na2CO3 solution is
- 0.2M
- 0.5M
- 0.05M
- 0.005M
- 0.01M
(iii) Which of the following compounds does Not belong to the Alkane homologous series
- C2H4
- CH4
- CsH12
- C3H8
- C4H10
(iv) The reaction between hydrogen and Iodine is represented by H2(g) + I2(g)![]() 2HI(g)
2HI(g) ![]() H=-xKJ/mol The reaction is
H=-xKJ/mol The reaction is
- Endothermic reaction
- Neutralization reaction
- Displacement reaction
- Exothermic reaction
- Decomposition reaction
(v) Chlorine gas was bubbled through distilled water. Then the distilled water turned pale yellow. Which of the following equations correctly show the reaction that took place?
- Cl2(g) + H2O(I) Cl2(g) +H2(g)
- Cl2(g) + H2O(I) HCl(aq) + HCIO(aq)
- 2Cl2(g) + H2O(I) 4Cl(aq) + O2(g)
- Cl2 + 2H2O(I) HOCl2 + H2O(I)
- Cl2(g) + H2O(I) HCl(aq) + H2O(I)
(vi) The following particles forms the nucleus of an atom
- Proton only
- Neutron and electron and Neutron
- Proton and Electron
- Proton and Electron
- Neutron and proton
(vii) One of the following apparatus is used to measure fixed volume of liquid
- Burette
- Pipette
- Conical flask
- Measuring cylinder
- Volumetric flask
(viii) A newspaper article claimed that carbon dioxide is formed as follows
- During respiration
- When calcium carbonate reacts with dilute HCI
- When methane burns completely in air. Which statement is correct
- 1, 2 and 3
- 1 and 2
- 1 and 3
- 2 and 3
- 1 and 2
(ix) Consider the following fuel which are used for different purpose
- Coal
- Firewood
- Petrol
- Charcoal
Which fuel originates from fossils?
- 1 and 3
- 2 and 4
- 1 and 4
- 2 and 3
- 3 and 4
(x) The following is an example of organic acid
- Hydrochloric acid
- Phosphoric acid citric acid
- Citric acid
- Nitric acid
- Carbonic acid
2. Match the descriptions in List A with the corresponding classes of fire in List B by writing the letter of the correct response beside the item number in the answer booklet provide.
| LIST A | LIST B |
|
|
SECTION B.(70 MARKS)
Answer all questions from this section.
3. (a)Give three (3) ways in which environmental destruction is likely to occur during charcoal production.
(b)A form one student was asked to separate different mixtures in the laboratory. What methods used to separate the following components of mixture?
- Sand and Iron fillings
- Oil from nuts
- Iodine and sand
- Kerosene and water
4. Consider the following part of periodic table then use it to answer the questions that follows
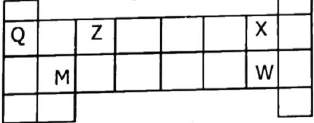 |
- Name the elements represented by letters X, Q and Z
- (i) Write the electronic configuration of M and W
(ii) Which letter represent one metal and which letter represent one non-metal
5. (a) Briefly explain the concept of homologous series and give two characteristics of it
(b)Give the IUPAC names of the following compounds.

6. Exactly 15.8cm3 of 0.3M HCl solution were enough to neutralize 23cm3 of sodium carbonate solution prepared by dissolving 29.46g of the hydrated sodium carbonate (Na2CO3, XH2O) in a litre of solution. Find the value of X which is the number of molecules of water of crystallization
7. (a)what is the difference between electrochemical equivalent and chemical equivalent.
(b)During electrolysis of AgNO3 and CUSO4 solutions 5.4 gram of silver were deposited at the cathode. Find mass of copper and volume of oxygen gas at STP
8. (a)By giving reasons explain the facts that
- Chlorine gas is collected by upward displacement of air
- Ammonia gas is not dried by concentrated sulphuric acid
- Sulphur trioxide (SO3) is not directly dissolved in water to form sulphuric acid
(b) By using electronic diagrams show bonding occurs in the following
- Nitrogen molecule (N2)
- Sodium fluoride
9. Form four students conducted experiment in the laboratory to synthesize oxygen gas, Sulphur trioxide and hydrogen gas. Their experimental results were tabulated as follows.
| Experiment | Reagents | Condition | Products |
| 1 | Potassium Chlorate |
| Potassium Chloride and oxygen gas |
| 2 | Zinc granule and dilute hydrochloric acid | Catalyst | Hydrogen gas |
| 3 | Sulphur dioxide and oxygen | Catalyst | Sulphur trioxide |
- Write word equations with corresponding chemical equations to summarize the reactions taking place in each of the experiment 1 to 3
- What are the names of catalysts used in experiment 1 and experiment 3
10. (a)What do you understand by the following concepts
- Fertilizer
- Manure
(b)A farmer wanted to enrich his farm with nitrogen. He went to the shop and found the following fertilizer with their respective price.
| Fertilizer Urea (NH2)2 CO | Price 40,000/= Per bag |
| Ammonium nitrate NH4NO3 | 40,000/= Per bag |
11. (a)(i)How many chlorine molecules are present in 30cm3 of chlorine gas at STP?
(iii)What are the condition for a substance to be called matter?
(b)28 gram of Iron reacted as follows
2Fe(s) + 3Cl2(q) → 2FeCl3(aq)
Calculate volume of chlorine gas at STP
12. (a)A form III student performed two different experiments
Experiment I: She dissolved one tea spoon of sugar in cold water.
Experiment II: She dissolved one tea spoon of sugar in hot water.
By using the concept of collision theory explain in which experiment the sugar dissolved more quickly?
(b)You are provided with the following equilibrium reaction
N2(g) + 3H2(g) ![]() 2NH3(g) + heat
2NH3(g) + heat
What happens to the production of ammonia if
- Pressure increased
- The equilibrium system is cooled
- Hydrogen is removed
- Nitrogen is added
SECTION C (15 MARKS)
Answer one (1) question from this section
13. Most of areas in Dar es Salaam have a problem of water hardness which affect much their daily life activities. As an expert explain how can help them to solve the problem.
14. (a)What are the four (4) stages for extraction of moderate reactive metals is termed as reduction process. Explain why?
(b)Extraction of metals is termed as reduction process. Explain why?
(c)In certain areas iron can be extracted through blast furnace which involve different temperatures such as 1000°C, 750°C and 250°C in different stages.
- What is a chief ore in extraction of iron
- Write equations taking place in each of the named temperatures at what temperature reduction takes place
- At what temperature reduction takes place.
- What are the importance of coke, hot air and waste gases in the process above?
(d) What are the two (2) environmental effects caused by extraction of metals?
FORM FOUR CHEMISTRY EXAM SERIES 59
FORM FOUR CHEMISTRY EXAM SERIES 59
PRESIDENT'S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
KILIMANJARO REGIONAL COMMISSIONER'S OFFICE
FORM FOUR MOCK EXAMINATION
CHEMISTRY 1 CODE: 032/1
TIME: 3 HOURS 29/06/2022
INSTRUCTIONS
1. This paper consists of sections A, B and C with a total of fourteen (14) questions.
2. Answer all questions in section A and B and one (1) question from section C.
3. Sections A and C carry fifteen (15) marks each and section B carries seventy (70) marks.
4. Cellular phones and any unauthorized materials are not allowed in the examination room.
5. Write your Examination Number on every page of your answer booklet(s)
6. The following constants may be used.
- Atomic masses: H=l, C= 12, K=39, S= 32, 0=16, N=14
- Avogadro's number = 6.02 x 1023
- GMV at s.t.p 22.4 dm
- 1 Faraday = 96,500 coulombs.
- Standard pressure 760 mmHg
- Standard temperature = 273 K.
- 1 litre = 1 dm3=1000cm3
SECTION A (15 Mario)
Answer all questions in this section.
1. For each of the item (i)-(x), choose the correct answer from among the given alternatives and write its letter beside the item number in the answer sheet provided.
(i) Access to safety equipment should never be blocked by any object because
- It is just a simple law
- It is important to reach safety equipment quickly in case of an accident
- The equipment is used every day
- There must be space for people to move around in the laboratory
- The equipment used to avoid accident in the laboratory
(ii) What should be done if the results obtained from an experiment do not support the hypothesis?
- The results should be left out
- A new problem should be identified
- The experiment should be changed
- Ideas for further testing to find a solution should be given
- The hypothesis should be accepted
(iii) Water exists in three forms, which are solid, liquid and vapour. Which among of the following are examples of liquid form of water
- Rain, snow and hail
- Dew, rain and ice
- Mist, steam and clouds
- Rain, hail, ice
- Rain, mist, and dew
(iv) Wind is a promising prospect source of energy in Tanzania because
- It cannot be seen easily
- It has high energy value
- Three are no chemical reactions involve in harvesting wind power
- It is renewable
- Does not produce harmful gases that pollute environment
(v) The reaction between potassium metal and water is REDOX reaction 2K(s)+2H2O(l) → 2KOH(aq) + H2(g)
Which of the following statement is true about this reaction?
- The element hydrogen is oxidised
- The element potassium is reduced
- Potassium is the reducing agent
- Hydrogen is the reducing agent
- Water is both oxidised and reducing agent
(vi)The number of molecules of oxygen gas of volume 4,480cm measured at STP is:
- 2 x 1023
- 6.02 x 1023
- 1.204 x 1023
- 22.4 x 1023
- 3.0 x 1022
(vii) Emulsion is the mixture of liquids that do not completely mix with each other.
Which of the following set represent emulsion?
- Butter, lotion, toothpaste and milk
- Milk, gel, foam, sponge
- Cream, gel, lotion and paint
- Sponge, milk, smoke, insecticides
- Smoke, milk, paint and body spray
(viii) The formulae below represent some chemical substances. Which formula represent a substance that does not contribute to global warming
- H2
- CH4
- CO2
- N2O
- SO2
(ix) Domestic utensils made up of iron do rust as a result of the presence of
- Air and fire
- Water and oil
- Air and water
- Air and oil
- Water and fire
(x) Ammonia is a basic gas that can be obtained by reacting ammonium salt and calcium hydroxide. Which of the following can be used as a drying agent of ammonia?
- Concentrated sulphuric acid
- Calcium chloride
- Lime water
- Calcium oxide
- Calcium carbonate
2. Match the properties of element in List A with the respective element in List B by; writing the letter of the correct response besides the item number in the answer sheet (s) provided.
| LIST A | LIST B |
| (i) A black solid element which burns with reddish glow giving off colorless gas which is slightly acidic (ii) Silvery white metal which burns with golden yellow flame giving an oxide which is basic in nature (iii)A yellow element in color which burns with blue flame giving colorless gas which is strong acidic in nature (iv) A shinning white strip metal burns with dazzling white flame giving an oxide which is slightly basic in nature (v) A silvery white metal which burns with brick red giving off oxide which is white solid |
|
SECTION B (70 Marks)
Answer an questions in this section
3. (a) The Modem periodic law is based on modification of Mendeleev periodic law. Explain how the two theories differ from each other.
(b) Comment on the following statement
(i) Lithium has large size than Beryllium
(ii) Sodium is smaller than potassium
(c) Give any four ions whose electronic configuration resemble to that of Neon. (07 Marks)
4. (a) A gaseous compound consists of 86% Carbon and 14% Hydrogen by mass. At S.T.P, 3.2dm3 of the compound had mass of 6g.
(i) Calculate its molecular formula
(ii)Give the IUPAC name of the compound
(b) Most of the apparatus in the laboratory are made up of glass materials. Support this statement by giving ant two (2) reasons. (07 Marks)
5. (a) Magnesium burns in a carbon dioxide living white solid and black solids sparks. The white solid dissolves in nitric acid leaving colorless solution R.
i. Write a balance equation for the reaction that gives the white solid.
ii. Identify solution R.
(b) In three points differentiate solution from suspension (07 Marks)
6. Msuya was given two gas jars, one containing gas "X" and another one containing gas "Z". Gas "X" is used prepare water gas in the industry and gas "Z" is used treating sewage plants.
(a) Identify those two gases
(b) (b)With reasons give the method used to collect gas "Z" in the laboratory
(c) Give three uses of gas "X" in our daily life (07 Marks)
7. A piece of marble chips (calcium carbonate) was placed in a beaker containing an excess of dilute hydrochloric acid standing on a reading balance. The mass of beaker and its contents was recorded after every two minutes as shown in the table below
| Time (min) | 0 | 2 | 4 | 6 | 8 | 10 | 12 | 14 |
| Mass (g) | 126.44 | 126.31 | 126.19 | 126.09 | 126.03 | 126.00 | 126.00 | 126.00 |
i. Why was there a loss of mass?
ii. Write the equation for the reaction
iii. State three different ways in which the reaction could have been more rapid
iv. Why did the mass remain constant after ten (10) minutes? (07 Marks)
8. (a) When an acid is reacted with base, it forms salt and water. Using your knowledge of chemistry, explain how will you apply this reaction in your daily life? Give any four points.
(b) Insoluble salts are the salt that does not dissolve in water. Name any three examples of salts that are insoluble in water. (07 Marks)
9. 16.8g of impure potassium hydroxide was dissolved in distilled water to make 1000mls. 20mls of this solution required 28mls of 0.07M sulphuric acid to react. Calculate:
(a) Molarity of KOH
(b) Mass Concentration of pure KOH (07 Marks)
10.During the manufacture of sulphuric acid, Sulphur trioxide gas is dissolved in concentrated sulphuric acid instead of dissolving it directly in water.
(a) Explain why? Write balanced equation of dissolution of the gas in the acid.
(b) Write a balanced chemical equation for each of the following
(i) Sulphuric acid reacting as an acid
(ii) Sulphuric acid reacting as an oxidizing agent (07 Marks)
11. (a) Mrs. Janeth bought all necessary building materials as he wanted to build modern and expensive house for his mother, four months later he found out that almost all nails and Iron sheets have been rusted as she ignored leakage and small holes in the storage room, unfortunately he has no extra money to buy new nails and Iron sheets. As a chemistry student what will you advice Mrs. Janeth?
(b) Suppose you are in a bus traveling from Moshito Rombo for annual holiday, a bus driver went to the petrol station to fill fuel but he forgets to switch off the bus engine, this caused explosion and fire started. As a form four students:
(i) Will you be able to use fire extinguisher to fight against fire?
(ii) Which type of extinguisher will be suitable for you to use? Give reason of your choice (07 Marks)
12. (a) A farmer was cultivated crops every year but he produced low yields because the farm had low soil pH. As a chemists advice the farmers three methods used in modifications of soil pH.
(b) Differentiate soil fertility from soil productivity (07 Marks)
SECTION C.
Answer one (1) question from this section.
13. (a)Eutrophication is the excessive growth of aquatic plants and algae in water bodies
(i) Give two causes of eutrophication
(ii) Explain the effects of eutrophication
(b) Explain how industrial workers can be protected against harmful effects of the chemicals.
14. (a) With the aid of equations briefly explain the term substitution reaction as applied in organic Chemistry.
(b) With the use of chemical tests differentiate between saturated hydrocarbons and unsaturated hydrocarbons
(c) How can you convert ethane to ethane? Use equation.
(d) Write all isomers of Pentene
FORM FOUR CHEMISTRY EXAM SERIES 21
FORM FOUR CHEMISTRY EXAM SERIES 21
THE UNITED REPUBLIC OF TANZANIA PRESIDENT’S OFFICE, REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
LUDEWA DISTRICT COUNCIL FORM FOUR MOCK EXAMINATION JUNE 2022.
032/1 CHEMISTRY 1
(For both school and private candidates)
Time 3 Hours 8th/06/2022
INSTRUCTIONS
1. This paper consists of section A, B & c with a total of fourteen (14) questions.
2. Answer all questions in section A & B and one (1) question from section C.
3. Cellular phones and any unauthorized materials are not allowed in the examination room.
4. Write your examination number on every page of your answer booklet(s).
5. The following constants may be used
Atomic masses: H=1, O=16, S=32, Ca=40, Zn=65, Cl=35.5, Na=23, C=12, Ag=108
Avogadro’s number = 6.02 x 1023 GMV at s.t.p = 22.4dm3
1 faraday = 96,500 coulombs
1 litre = 1dm3 = 1000cm3
SECTION A (15Marks)
Answer all questions in this section
1.For each of the items (i) – (x), choose the correct answer from among the given alternatives and write its letter beside the item number in the answer booklet provided.
i. Mr KALOBERO performed an electrolytic experiment on Calcium chloride.What number of faradays of electricity is required to deposit 4g of calcium from molten calcium chloride?
- 0.1
- 0.2
- 0.4
- 0.3
- 0.7
ii. Miss Mfwimi took methane and did substitution reaction with excess chlorine. What is the final product?
- Chloromethane
- Dichloromethane
- Trichloromethane
- Tetracloromethane
- Monochloromethane
iii. Mrs. Chakubanga mixed different metals uniformly. Which name is given to the uniform mixture of metals?
- Compounds
- Alloys
- Ores
- Salts
- Mixtures
iv. Dr Mbogo did a certain reaction. The reaction was between iodine and hydrogen as represented by the following equations
 |
The reaction is:-
- An endothermic reaction
- A displacement reaction reaction
- A neutralization reaction
- A thermal decomposition
- An exothermic reaction
v. A student teacher was in the class teaching how to write electronic configuration of different elements. The electronic configuration of this element Cl- is
- 2:8:7
- 2:8:8
- 2:8:8:1
- 2:8:8:2
- 2:8:6
vi. When Mr Mwendakwao took Glucose and dissolved in water to form a uniform mixture.
- water is solution, glucose is solvent and the product is solute
- water is solute, glucose is solvent and the product is solution
- water is solvent, glucose is solute and the product is solution
- water and glucose forms immiscible mixture
- water and glucose form emulsion
vii. Tell to Mr. Kitwanga, left white anhydrous copper (II) Sulphate in air turned blue. The reason why white anhydrous copper (II) Sulphate turns blue when exposed in Atmosphere is that it
- Absorbs water vapour
- Reacts with oxygen
- Reacts with carbon dioxide
- Become dry
- Release water to the Atmosphere
viii. In order for Mr. Kingunge to produce the greatest amount of hydrogen in a short time, one gram of magnesium ribbon should react with
- 10cm3 of 0.5M Sulphuric acid
- 40cm3 of 0.5M acetic acid solution
- 40cm3 of 0.5 Sulphuric acid solution
- 20cm3 of 1M Sulphuric acid solution
- 20cm3 of 1M acetic acid solution
ix. Mr. Kigwangala was just complaining about the increases of pollutant gases in the atmosphere causes the increase in temperature. but some gases cause acid were not present to Kigwangala’s complain. Make him understand what are the acidic oxides gases.
- Carbon dioxide, carbon monoxide and Sulphur dioxide.
- Sulphur trioxide, Nitrogen dioxide and Nitrogen monoxide
- Carbon dioxide, Sulphur dioxide and dinitrogen oxide
- Sulphur trioxide, carbon dioxide and Nitrogen dioxide
- Carbon monoxide, nitrogen oxide and Sulphur dioxide.
x. Mrs.Tupatupa spilled out concentrate acid on the floor. Which action should be taken immediately after concentrated Sulphuric acid is spilled on the skin?
- It should be rinsed off with large quantities of running water.
- It should be neutralized with concentrated NaOH
- The affected area should be wrapped tightly and shown to a medical health provider
- It should be Neutralize with solid CaCO3
- It should be neutralized with concentrated KOH
2 Match the items in List A with the response in List B by writing the letter of the corresponding responses beside the item number.
| LIST A | LIST B |
| i. Biogas ii. Biomass iii. Natural gas iv. Producer gas v. Water gas |
|
SECTION B (70Marks)
Answer all questions in this section
3. (a)(i) A student from pre form one class asked you teach him the real meaning of Laboratory. What was you explained so as to make him understand the meaning of laboratory?
(ii) Mr. Kiseyeye wanted to prepare an oxygen. What are two main compound do Mr. Kiseyeye used in preparation oxygen gas in laboratory.
(ii)You as a student from form four class what do you think Why hydrogen gas is collected by downward displacement air method?
(b)Mr Kadinya don’t understand how to complete and balance chemical reactions. Help mr Kadinya to Complete and balance the following chemical reaction
i. NaOH(ag) + HCl(ag) →
ii. NH4Cl(ag) +NaOH(ag) →
iii. Ca(OH)2(ag) +H2SO→
iv. Mg(s) +HNO3(ag) →
4. Element R having atomic number 20 combines with element S having atomic number 17 to form a certain compound
a) Write the formula of the compound and state the type of bond formed in the compound
b) Give any three properties of the compound formed in 7(a) above
5. .As a competent chemistry student chosen to present the topic of mole concept and related calculations. Show how will you present the following calculations
i. How many chlorine molecules are in 20cm3 of chlorine gas at s.t.p.
ii) Calculate number of ions present in 5g of copper (II) Nitrate
6. a) Muumini is a student from Katosani secondary school, she is not able to State Le Chatellier’s principle, Help her.
b) In the industrial preparation of Sulphur trioxide mr Atieno yo established equilibrium between Sulphur dioxide and oxygen gas as follows:
2SO2(g) + O2(g) ![]() 2SO3(g)?H= - 94.9KJ/mol.
2SO3(g)?H= - 94.9KJ/mol.
i. How would you adjust temperature and pressure to maximize the proportion of the product at equilibrium?
ii. Why is it unfavorable to work with very high pressure and very low temperature in the contact process?
iii. What catalyst is used to speed up the rate of formation of Sulphur trioxide before attaining the equilibrium?
7. a) You are tasked to explain the meaning of Molecular formula in a certain presentation that will be held next year. Present it now.
b) You are provided with a compound composed of 22.2% Zinc, 11.6% Sulphur, 22.3% oxygen and the rest percentage is water of crystallization. Calculate the molecular formula of the compound if its molecular mass is 283.
8. In one of titration experiment 25cm3 of hydrated sodium carbonate Na2CO3.XH2O were titrated against 20cm3 of 0.25M hydrochloric acid. The solution of sodium carbonate was made by dissolving 7.15g of the compound in 250cm3 of distilled water.
Use the above data to answer the following questions
a) Write down a balanced equation to represent the acid base reaction
b) i. Find the morality of the an hydrated sodium carbonate ii. Calculate the value of X in the hydrated sodium carbonate.
9. a) Mr Mapulishi tasked to prepare all requirements for extraction of sodium.Describe the use of each of the following during extraction of Sodium
i. Calcium Chloride ii. Graphite anode
iii. Steel gauze
b) .Why sodium is collected by upwards in the downs cell
c) Write electrodes reaction in downs cell during extraction of Sodium
10. a) When I was in an interview at Feza boys’ secondary school, one student asked me to State Faraday’s Laws of Electrolysis. If you were me answer such question.
b) Dilute silver nitrate solution was decomposed by the passage of electric current through it. What mass of silver and what volume of oxygen (Measured at s.t.p) would be liberated in electrolysis by 9650 coulombs of Electricity?
11. Mr Kalubandika wanted to know some chemistry pertaining concepts. Help Mr Kalubandika to answer the following conceptual questions.
a) In which other areas do we find the warning signs out of laboratory (give four point)
b) Explain how measurements of volume differ when using measuring cylinder and burette
c) It is recommend that laboratory apparatus should be properly washed or wiped after use, explain the significance for this when
i. Measuring volume of liquids
ii. Measuring mass of substance
12. a) Mr Mwakatumbula don’t understand the physical properties of water. Teach him by giving three points the main physical properties of water and show the usefulness of each property.
b) Mr Kadinya said that Electrolysis is applied by many areas on the earth. State four industrial application of electrolysis
SECTION C (15 MARKS)
13. We have coal at Kiwira in Mbeya region. Authorities in the government have allowed use of coal for domestic and industrial purpose. What warming can you raise concerning likely effects? Give five points
14. a). Explain the mechanism of electrolysis
b). briefly explain three application of electrolysis in our daily life
FORM FOUR CHEMISTRY EXAM SERIES 15
FORM FOUR CHEMISTRY EXAM SERIES 15
THE UNITED REPUBLIC OF TANZANIA PRESIDENT’S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
MWANZA CITY JOINT EXAMINATION (NYEGEZI, BUTIMBA AND MKUYUNI WARDS)
FORM FOUR PRE - MOCK EXAMINATION-2021
CHEMISTRY
Time 3:00 Hrs MAY, 2021
Instructions
- This paper consists of section A, B & c with a total of fourteen (14) questions.
- Answer all questions in section A & B and one (1) question from section C.
- Cellular phones and any unauthorized materials are not allowed in the examination room.
- Write your examination number on every page of your answer booklet(s).
- The following constants may be used
- Atomic masses: H=1, O=16, S=32, Ca=40, Zn=65, Cl=35.5, Na=23, C=12, Ag=108
- Avogadro’s number = 6.02 x 1023
- GMV at s.t.p = 22.4dm3
- 1 faraday = 96,500 coulombs
- 1 litre = 1dm3 = 1000cm3
SECTION A (15Marks)
Answer all questions in this section
1. For each of the items (i) – (x), choose the correct answer from among the given alternatives and write its letter beside the item number in the answer booklet provided.
- What number of faradays of electricity is required to deposit 4g of calcium from molten calcium chloride?
- 0.1
- 0.2
- 0.4
- 0.3
- 0.7
- When methane undergoes substitution reaction with excess chlorine. What is the final product?
- Chloromethane
- Dichloromethane
- Trichloromethane
- Tetracloromethane
- Monochloromethane
- Which name is given to the uniform mixture of metals?
- Compounds
- Alloys
- Ores
- Salts
- Mixtures
- The reaction between iodine and hydrogen is represented by the following equations
I2(g) + H 2(g) ![]() 2HI (g)
2HI (g)![]()
The reaction is:-
- An endothermic reaction
- A displacement reaction
- A neutralization reaction
- A thermal decomposition reaction
- An exothermic reaction
- The electronic configuration of this element Cl- is
- 2:8:7
- 2:8:8
- 2:8:8:1
- 2:8:8:2
- 2:8:6
- Glucose dissolves in water to form a uniform mixture.
- water is solution, glucose is solvent and the product is solute
- water is solute, glucose is solvent and the product is solution
- water is solvent, glucose is solute and the product is solution
- water and glucose forms immiscible mixture
- water and glucose form emulsion
- The reason why white anhydrous copper (II) Sulphate turns blue when exposed in Atmosphere is that it
- Absorbs water vapour
- Reacts with oxygen
- Reacts with carbon dioxide
- Become dry
- Release water to the Atmosphere
- In order to produce the greatest amount of hydrogen in a short time, one gram of magnesium ribbon should react with
- 10cm3 of 0.5M Sulphuric acid
- 40cm3 of 0.5M acetic acid solution
- 40cm3 of 0.5 Sulphuric acid solution
- 20cm3 of 1M Sulphuric acid solution
- 20cm3 of 1M acetic acid solution
- Which of the following substances represent a group of acid oxides?
- Carbon dioxide, carbon monoxide and Sulphur dioxide.
- Sulphur trioxide, Nitrogen dioxide and Nitrogen monoxide
- Carbon dioxide, Sulphur dioxide and dinitrogen oxide
- Sulphur trioxide, carbon dioxide and Nitrogen dioxide
- Carbon monoxide, nitrogen oxide and sulphur dioxide.
- Which action should be taken immediately after concentrated sulphuric acid is spilled on the skin?
- It should be rinsed off with large quantities of running water.
- It should be neutralized with concentrated NaOH
- The affected area should be wrapped tightly and shown to a medical health provider
- It should be Neutralize with solid CaCO3
- It should be neutralized with concentrated KOH
- Match the items in List A with the response in List B by writing the letter of the corresponding responses beside the item number.
| LIST A | LIST B |
| (i) Biogas (ii) Biomass (iii) Natural gas (iv) Producer gas (v) Water gas |
|
SECTION B (70Marks)
Answer all questions in this section
3. (a)
- What is laboratory?
- Name two main compound used in preparation oxygen gas in laboratory
- Why hydrogen gas is collected by downward displacement method?
(b) Complete and balance the following chemical reaction
- NaOH(ag) + HCl(ag)

- NH4Cl(ag) +NaOH(ag)

- Ca(OH)2(ag) +H2SO4(ag)

- Mg(s) +HNO3(ag)

4. Element R having atomic number 20 combines with element S having atomic number 17 to form a certain compound
(a)Write the formula of the compound and state the type of bond formed in the compound
(b) Give any three properties of the compound formed in 7(a) above
5. a) How many chlorine molecules are in 20cm3 of chlorine gas at s.t.p.
b) Calculate number of ions present in 5g of copper (II) Nitrate
6. a) State Le Chatellier’s principle.
b) In the industrial preparation of Sulphur trioxide equilibrium is established between Sulphur dioxide and oxygen gas as follows:
2SO2(g) + O2(g) ![]() O3(g)?H= - 94.9KJ/mol.
O3(g)?H= - 94.9KJ/mol.
- How would you adjust temperature and pressure to maximize the proportion of the product at equilibrium?
- Why is it unfavorable to work with very high pressure and very low temperature in the contact process?
- What catalyst is used to speed up the rate of formation of Sulphur trioxide before attaining the equilibrium?
7.(a) What is molecular formula?
(b) You are provided with a compound composed of 22.2% Zinc, 11.6% Sulphur, 22.3% oxygen and the rest percentage is water of crystallization. Calculate the molecular formula of the compound if its molecular mass is 283.
8. In one titration experiment 25cm3 of hydrated sodium carbonate Na2CO3.XH2O were titrated against 20cm3 of 0.25M hydrochloric acid. The solution of sodium carbonate was made by dissolving 7.15g of the compound in 250cm3 of distilled water.
Use the above data to answer the following questions
- Write down a balanced equation to represent the acid base reaction
- i. Find the morality of the an hydrated sodium carbonate
ii. Calculate the value of X in the hydrated sodium carbonate.
9. (a)Describe the use of each of the following during extraction of Sodium
- Calcium Chloride
- Graphite anode
- Steel gauze
(b).Why sodium is collected by upwards in the downs cell
c). Write electrodes reaction in downs cell during extraction of Sodium
10. (a) State Faraday’s Laws of Electrolysis
(b) Dilute silver nitrate solution was decomposed by the passage of electric current through it. What mass of silver and what volume of oxygen (Measured at s.t.p) would be liberated in electrolysis by 9650 coulombs of Electricity?
11. (a) In which other areas do we find the warning signs out of laboratory (give four point)
(b) Explain how measurements of volume differ when using measuring cylinder and burette
(c) It is recommend that laboratory apparatus should be properly washed or wiped after use, explain the significance for this when
- Measuring volume of liquids
- Measuring mass of substance
12. (a) State three main physical properties of water and show the usefulness of each property
(b) State four industrial application of electrolysis
SECTION C (15 MARKS)
Answer one (1) question in this section
13. (a) How can you tell water is polluted? Give two ways
(b) We have coal at Kiwira in Mbeya region. Authorities in the government have allowed use of coal for domestic and industrial purpose. What warming can you raise concerning likely effects? Give five points
14. (a) Define the following terms
- functional group
- homologous series
- isomerism
(b) Write down the molecular structure and the IUPAC names of the isomer whose molecular structure is C4H10
(c) By naming the reagents, stating the conditions whenever possible using a balances equation describe how ethane could be converted into
- Ethane
- Chloroethane
- 1,2-dibromoethane
- Ethanol
FORM FOUR CHEMISTRY EXAM SERIES 69
FORM FOUR CHEMISTRY EXAM SERIES 69
THE PRESIDENT’S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
KILOSA DISTRICT COUNCIL MIKUMI (MAU)
FORM FOUR PRE - MOCK JOINT EXAMINATION
032/1 CHEMISTRY 1
(For School Candidates Only)
TIME: 3:00 Hours Friday March 19, 2021
Instructions
1. This paper consists of sections A, B and C with a total of fourteen (14) questions.
2. Answer all questions in sections A and B and one (1) question from section C.
3. Non-programmable calculators may be allowed.
4. Cellular phones and any unauthorized materials are not allowed in the examination room.
5. Write your Examination Number on every page of your answer booklet(s).
6. The following constants may be used: Atomic masses:
- Atomic masses; H = 1, O = 16, Na = 23, C = 12, Cl = 35.5, N =14, Zn = 65, S = 32, Ca = 40, Fe = 56, Cu = 64, Al = 27 .
- GMV at s. t. p = 22.4dm³
- Avogadro’s number = 6.02 x 10²³
- 1Faraday = 96500coulombs
- 1Litre = 1dm³ = 1000cm3
SECTION A (15 MARKS)
1. For each of the items (i – x) choose the correct answer from the given alternatives and write it’s letter in the booklet provided
(i) A substance which absorb water from the atmosphere and form a solution is called
- Efflorescent
- hygroscopic
- deliquescent
- amphoteric
(ii) Technicians prefer to use blue flame in welding because
- It is bright and non soot
- it is light and non soot
- it is very hot and non soot
- it is not expensive
(iii) Chlorine ion Cl? differ from chlorine atom because it has
- More proton
- less proton
- more electron
- less electrons
(iv) AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNo3(aq). This a good example of
- Neutralization reaction
- double decomposion reaction
- Redox reaction
- synthesis reaction
(v) The volume of 0.2M of H2SO4 acid required to neutralize completely 25.00cm³ of 0.05M KOH is
- 0.626cm³
- 3.125cm³
- 12.500cm³
- 6.315cm³
(vi) An electric current was passed through concentrated hydrochloric acid using carbon electrodes. The substance liberated at the anode was
- copper
- Chlorine
- oxygen
- hydrogen.
(vii) An example of a homologous series is
- methane ,ethane, propane
- propane, butane, pentyne
- ethane, propene, butyne
- ethene, propane, butane.
(viii)A covalent bond is formed when
- ammonia is formed
- potassium and oxygen combine
- a metal combines with non metal
- an atom loses an electron .
(ix) The empirical formula of a certain compound is CH3.Its molar mass is 30g/mol , what will be its molecular formula
- C2H6
- CH4
- C2H8
- C2H4
(x) …… is the general terms used to explain a mixture of different metals
- Amphoteric
- allotrope
- isotope
- Alloy
2. Match the item in LIST” A” with the correct response in LIST” B”
| LIST A | LIST B |
| (i) Methyl orange indicator (ii) Calcium hydroxide (iii) pH 2 (iv) Neutralization reaction (v) Sodium hydrogen sulphate |
|
SECTION B (70 MARKS)
3. (a) Write balanced equation of
(i) Sodium hydroxide react with sulphuric acid
(ii) Calcium carbonate decomposed by heat
(b) (i) With aid of a balanced chemical equation name the products formed when nitrates of potassium and zinc decompose by heat
(ii) Suggest why nitrates of zinc and potassium behave differently on heating
4. (a) (i) People suffering from heart burn usually use wood ashes for relief. Mention chacteritics which makes the ashes to be used for heart burn relief.
(ii) Give four compounds found in the laboratory which show the same characteristic as ashes.
(b) How many ions are there in 6.82g of Al2(SO4)3
5. (a) Copy and complete the following table
| Element | A | B | C | D |
| Number of proton | | | | 16 |
| Number of neutron | 6 | 12 | | |
| Mass number | | | 39 | 32 |
| Electronic configuration | | 2:8:1 | | |
(b) (i) Write balanced chemical equation for reaction between B and D
(ii) Write the molecular formula of a compound formed when C combine with D
6. A students tested four sample of water each 10cm³ from different areas at Malawi village by shaking with tree drops of soap solution. The experiment was repeated a second time by boiling each sample of water (10cm³) with three drops of soap solution. The observation were recorded as shown in the table below
| Sample | Observation with soap solution | Observation for boiled sample with soap |
| A | No lather | Lather |
| B | Lather | Lather |
| C | Lather | Lather |
| D | No lather | No lather |
a) Which sample contains only temporary hard water? Give reason
b) Which sample contain permanent hard water? Give reason
c) Name the chemical substances that would be the causes of hardness in sample “A”
d) Write the chemical equation for removing hardness in
(i) Sample “A”
(ii) Sample “D”
7. (a) (i) State faraday’s first law of electrolysis
(ii) Using the law stated above, derive mathematical expression of faraday’s first law of electrolysis
(b) How many moles of electrons will be transferred if Zinc metal is produced by a current of 14.23A supplied for 8.0 HRS.
8. 5.3g of X2CO3 was dissolved in water to make 0.5 litre of a solution. 25cm³ 0f this solution required 50.0cm³ of 0.1M HCl for complete neutralization.
a) Write the balanced chemical equation for the reaction
b) Calculate the concentration of X2CO3 in mol/dm3
c) Calculate the relative molecular mass of X2CO3
d) Calculate the relative atomic mass of X
e) What is the name and symbol of element X
9. (a) Give two example in each of the following
(i) Solid fuel (ii) Gaseous fuel
(b) The reaction which produces methanol from carbon monoxide and hydrogen is represented by the equation
.The reaction is carried out at high pressure to give a better yield of methanol
i) Explain why increase in pressure gives a better yield of methanol.
ii) The value of 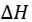 is negative . What does this tell about the reaction
is negative . What does this tell about the reaction
iii)With reason state whether a high temperature or low temperature will give a better yield of methanol.
10. Draw the well labeled diagram of laboratory preparation of oxygen using the mixture of potassium chlorate and manganese
(iv)oxide
11. (a) Define the oxidation state
(b) Calculate the oxidation state of the underlined element in the following compounds
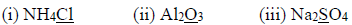
12. (a) Define the following
i)A homologous series
ii) Hydrocarborns iii) Isomerism
(b) Write the structure formulae of all possible isomers of hydrocarbon whose formula is C4H10 and give the IUPAC names.
SECTION C (15 MARKS)
Answer only one (1) question from this section
13. With the aid of the balanced chemical equations explain the processes that occurs in the blast furnace during extraction of iron.
14. Environment support lives of all organism. Its pollution has led to some major catastrophic effects. Describe water pollution by analyzing it’s causes, effect and the protective and remedial measures be taken.
FORM FOUR CHEMISTRY EXAM SERIES 52
FORM FOUR CHEMISTRY EXAM SERIES 52
PRESIDENT’S OFFICE REGIONAL ADMINISTRATION AND LOCAL GOVERNMENT
FORM IV MOCK EXAMINATION 2020 DODOMA REGION
032/1 CHEMISTRY 1
Time: 3 Hours Thursday, 6th August 2020 a.m.
Instructions
1.This paper consists of three sections A, B and C with total of fourteen (14) questions.
2.Answer all questions from section A and B and one (1) question from section C.
3.Except for diagrams that must be drawn in pencil, all writing should be done using a blue or black pen.
4.Calculators and cellular phones are not allowed in the examination room 5. Write you Examination number on every page of your answer sheets/booklet(s)
6. The following constants may be used:
Atomic masses: H = 1, C = 12, N = 14, O = 16, Na = 23, S = 32, Cl = 35.5, Cu = 63.5 and Pb =207
Avogadro’s number = 6.02 x 1023mol-1
GMV at s.t.p = 22.4 dm3mol-1
1 Faraday = 96500 Coulombs
Standard temperature = 273 K
1 litre = 1 dm3 = 1000 cm3
SECTION A (15 Marks)
Answerall questions in this section
1. For each of the following items (i) – (x) choose the correct answer among the given alternatives and write its letter beside the item number in the answer sheet(s) provided
(i) Splints are among the first aid kit components. Which of the following sentences best describes its use?
A. Used for cutting bandages or piece of cloth
B. Used for holding pieces of bandages or cloth together
C. Used to support broken bones and is tied using bandages
D. Used for picking up and holding things
E. Used for cleaning wounds and make it heal faster
(ii) A simple proof that some chemical reactions take place in our bodies is that
A. We eat a balanced diet
B. Doctors tell us so in the hospitals
C. We occasionally fall sick
D. The food we eat or the drinks we take are quite different from the waste products from our bodies
E. There is no proof
(iii) The following reaction, 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g), is an example of a
A. Decomposition reaction
B. Combination reaction
C. Redox reaction
D. Neutralization reaction E. Esterification reaction
(iv) An aqueous solution turns red litmus solution blue. Excess addition of which of the following solution would reverse the change?
A. Baking powder
B. Lime
C. Aqueous ammonia
D. Vinegar
E. Quick lime
(v) Oils on treating with hydrogen in the presence of nickel catalyst form fats. This is an example of
A. Oxidation reaction
B. Addition reaction
C. Displacement reaction
D. Substitution reaction
E. Precipitation reaction
(vi) Three elements T, Q and R, are in the same period of the periodic table. The oxide of T is amphoteric, the oxide of Q is basic and the oxide of R is acidic. Which of the following shows the elements arranged in order of increasing atomic number?
A. T, Q, R
B. Q, T, R
C. Q, R, T
D. R, T, Q
E. T, R, Q
(vii) What happens when dilute sulphuric acid is poured on copper plate?
A. Zinc sulphate formed D.Copper chloride formed
B. Zinc chloride formed E.None of the above C. Copper sulphate formed
(viii) The oxidation state of chlorine in potassium chlorate is
A. + 1 C. + 5 E. + 6
B. + 7 D. – 1
(ix) Ammonia gas is collected by which method among the following?
A. Downward displacement of water
B. Upward delivery
C. Downward delivery
D. Upward displacement of air
E. Collected over water
(x) Ten gram of copper powder were placed in a sealed initiation tube containing air. The tube was heated very strongly for half an hour. At the end of the experiment
A. Water vapour appeared in the tube
B. Undergoes decomposition
C. The total mass of the tube and its contents decreased
D. Copper melted into a liquid copper form
E. Copper increased in mass and changed in colour
2. Match the items in List A with the responses in List B by writing the letter of the correct response beside the item number in the answer sheet or booklet provided.
| List A | List B |
| A. Water is used for extinguishing B. Oxygen and water/moisture C. Presence of Oxygen D. Fire triangle E. Presence of carbon dioxide F. Rusting G. Galvanization H. Class B fire I. Nitrogen |
SECTION B (70 Marks)
Answerall questions in this section
3. (a) The table below shows two brands of bottled water for drinking and the concentration of different mineral ions in each brand. Study the table and answer the questions below it.
| Composition in mg/Litre | Mineral | Na+ | Ca2+ | Mg2+ | Cl? | NO3? | SO42- | Fe2+ | F? |
| Uhai | 40.0 | 3.05 | 4.15 | 14.18 | 0.48 | 10.0 | 0 | 1.76 | |
| Dasani | 22.32 | 2.69 | 0.11 | 6.50 | 1.0 | 8.0 | 0 | 0.45 |
(i) Which brand of water is more hard? Explain.
(ii) State the benefit of having calcium ions in water;
(iii)Tap water is usually treated before being used. State what is added to perform that function.
(b) Hydrogen and phosphorus are non-metallic elements
(i) Which one between the two atoms is more electropositive?
(ii) Show your work clearly, write the chemical formula and the name of the compound formed when the two atoms combine.
4. One of the methods used for the preparation of oxygen is by the decomposition of hydrogen peroxide as shown in the reaction equation below
![]() 2(g)
2(g)
(a) (i) What is the purpose of MnO2 in the reaction?
(ii) What will happen to the rate of the reaction if MnO2 is not used in the above reaction?
(b) Briefly explain how the gas produced from the chemical reaction above is tested
5. (a) State the main raw materials and process involved in the manufacture of each of the following products
(i) Wood charcoal
(ii) Coke
(iii)Lamp black
(iv) Animal charcoal
(b) Name two main products formed when
(i) Hot concentrated sulphuric acid is added to sugar cane (ii) Hot concentrated sulphuric acid is added to ethanol
6. (a) Define the following terms
(i) Mole
(ii) Molar mass
(b) 112 dm3 of oxygen gas was collected at S.T.P when a sample of lead nitrate was completely decomposed by heat. Calculate the volume of nitrogen dioxide produced.
7. A gaseous compound consists of 86% carbon and 14% hydrogen by mass. At s.t.p., 3.2 dm3 of the compound had mass of 6 g. Calculate its molecular formula
8. (a) Study the following reaction equation N2(g) + 3H2(g)![]() 2NH3(g) ?H = - 46.2 kJmol-1
2NH3(g) ?H = - 46.2 kJmol-1
Use Le-Chatelier’s principle, suggest how you would use temperature and pressure to obtain the highest production of ammonia at equilibrium
(b) The formation of methanol from hydrogen and carbon monoxide can be represented by
CO(g) + 2H2(g) ![]() CH3OH ?H = 91 kJmol-1
CH3OH ?H = 91 kJmol-1
What mass of hydrogen would react to cause a heat change of 91 kJ
9. (a) Give the meaning of the following terms
(i) Soil pH (ii) Liming
(b) (i) Explain why sulphur and its compounds are removed from the fuel before they are burned
(ii) By using a reaction equation explain how propane differs from propene
10. (a) Outline any three uses of carbon dioxide gas in animals and plants
(b) Complete the following reactions
(i) CH2=CH2 + O2![]()
(ii) CaC2 + 2H2O ![]()
(iii)CH4 +Cl2 ![]()
(iv) C2H5OH +O2 ![]()
11. (a) (i) What is the aim of having fume chamber in the chemistry laboratory?
(ii) Why laboratory doors are open outward?
(b) State the use of the following
| Item | Use |
| (i) | Cotton wool |
| (ii) | Petroleum jelly |
| (iii) | Pain killers |
| (iv) | Bandages |
| (v) | Razor blade |
12. (a) With examples list four types of oxides
(b) Give any three types of salts
SECTION C (15 Marks)
Answer only one question in this section
13. Explain six (6) methods of preventing water pollution
14. In certain chemical plant that deals with the production of hydrogen gas, a certain chemist passes an electric current in series through copper (II) sulphate and sulphuric acid solutions. In both electrolytes, inert electrodes were used. If 8400 cm3 of hydrogen gas at S.T.P were produced for several hours, what volume of oxygen gas would also be produced? What mass of copper was produced?
FORM FOUR CHEMISTRY EXAM SERIES 10
FORM FOUR CHEMISTRY EXAM SERIES 10
Hub App
 For Call,Sms&WhatsApp: 255769929722 / 255754805256
For Call,Sms&WhatsApp: 255769929722 / 255754805256
 For Call,Sms&WhatsApp: 255769929722 / 255754805256
For Call,Sms&WhatsApp: 255769929722 / 255754805256







